Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
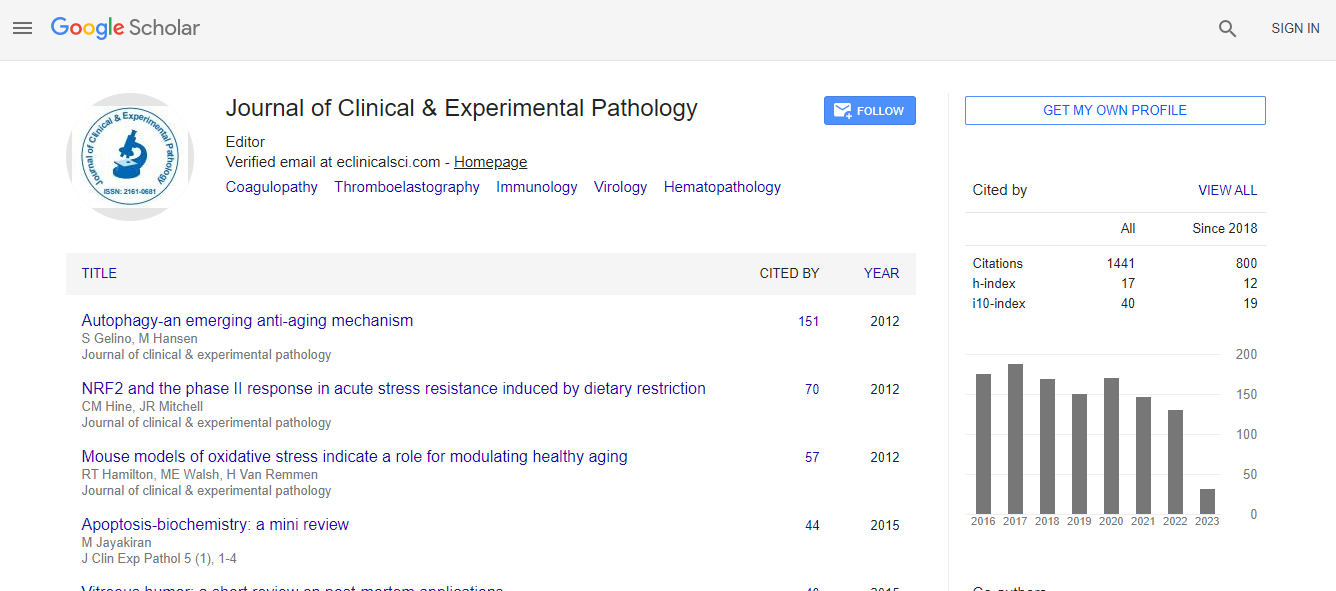
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Digital molecular pathology
14th Asia Pacific Pathology Congress
Ahmed A Yameny
Society of Pathological Biochemistry and Hematology, Egypt
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
The molecular pathology laboratory offers molecular diagnostic testing for a variety of clinical indications across the healthcare continuum, including testing for disease susceptibility, population screening, diagnosis, prognosis, therapeutic decision making and disease monitoring. Molecular Pathology (MP) is at the heart of modern diagnostics and translational research. It has become evident that, to advance in the translation of biomarker discovery into diagnostic and therapeutic application, the purpose of this article is to advance MP into Digital Molecular Pathology (DMP), depending on known biomarkers especially miRNA and proteomics, to resolve many of the demands in the clinical laboratory, as screening for cancer, endemic or rare virus infection and biological war. We would like to suggest an integrated model of DMP, from biomarkers to form two steps of diagnosis and two steps of treatment, the first step for fast diagnosis and fast therapy to stop spreading of disease, the second step for complete diagnosis and effectiveness treatment, many biomarkers were discovered so must divided into groups, DMP can allow facilities to outsource all or a portion of the demands data such as cloud computing, bioinformatics pipelines, variant data management and knowledge curation. Exchange of electronic molecular data allows laboratories to perform validation of rare diseases using foreign data, check the accuracy of their test results against benchmarks.Biography
Ahmed A Yameny is the Head of Society of Pathological Biochemistry and Hematology. He is Union Chief Medical Laboratory in Egypt. He was the Chairman for five international conferences of medical laboratory and two international conferences of the society. He is an Expert and Medical Laboratory Consultant. He is the Chief Editor of Journal of Bioscience and Applied Research. He has completed his BSc in Biochemistry from Alexandria University in Egypt and BSc in Biochemistry/ Chemistry from Tanta University in Egypt.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi