Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
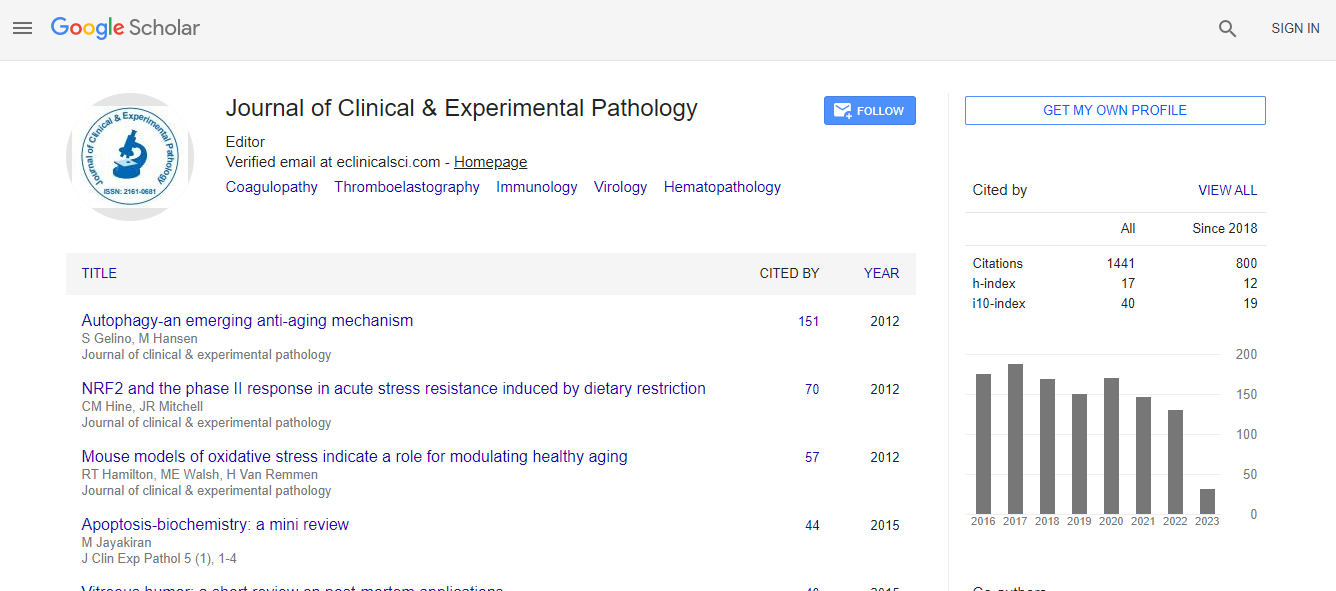
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Evaluation of Cepheid Xpert-Carba-R assay for the detection of carbapenemase-producing organisms
13th European Pathology Congress
Jaber Aslanzadeh
Hartford Health System, USA
Posters & Accepted Abstracts: J Clin Exp Pathol
Abstract
Background & Aim: Infections with carbapenem resistant microorganisms are associated with a high morbidity and mortality. Carbapenem resistance makes empirical and targeted treatment of infections challenging. Carbapenem resistance can arise by several mechanisms with the greatest emphasis on those bacteria that have acquired transferable carbapenemase genes such as KPC, OXA 48, IMP-1, VIM and NDM. The goal of this study was to compare the performance of the Cepheid Xpert-Carba-R assay to modify Hodge test (MHT) and carbapenem inactivation method (CIM). Method: MHT, CIM and Carba-R were performed on a panel of 24 isolates that included Acinetobacter baumannii (7), Klebsiella pneumoniae (9), Klebsiella oxytoca (1) Citrobacter freundii (1), E. coli (3), Morganella morganii (1) and Pseudomonas aeruginosa (2) as well as 18 carbapenem resistant organisms isolated from patients at Hartford Hospital [ E. coli (11), K. oxytoca (3), K. pneumoniae (7), P. aeruginosa (1) and Serratia marcescens (1)]. Antimicrobial susceptibility of the panel isolates revealed that all except for one A. baumannii, one E. coli and one K. pneumoniae were resistant or intermediate to one or more carbapenems. Similarly, antimicrobial susceptibility of the clinical isolates revealed that all isolates were resistant or intermediate to one or more carbapenems except for S. marcescens. Results: Of the 42 isolates, 21 isolates tested negative and 15 isolates tested positive for carbapenemase by all three tests. Two isolates (Acinetobacter baumannii) tested were positive by MHT, negative by CIM and CarbaR PCR and; one isolate (Pseudomonas aeruginosa) was tested positive by CIM, negative by MHT and CarbaR PCR. One isolate (Acinetobacter baumannii) tested positive by MHT and CIM, tested negative by CarbaR PCR and; one isolate (Acinetobacter baumannii) tested positive by CarbaR PCR (NDM) and MHT tested negative by CIM. One isolate (Klebsiella oxytoca) tested positive by CarbaR (NDM), tested negative by MHT and CIM. This was considered as a true positive missed by MHT and CIM. Conclusions: CarbaR detected all isolates with KPC, OXA 48, IMP-1, VIM and NDM carbapenemases. Both MHT and CIM were associated with false positive and false negative results. The turn-around time for CarbaR was less than 90 minutes compared to 24 hours for MHT and CIM.Biography
Jaber Aslanzadeh has completed his PhD at American Board of Medical Microbiology (ABMM). He is the Director of Clinical Microbiology at Hartford Hospital, USA. Currently, he is an Associate Professor of Laboratory Medicine at University of Connecticut School of Medicine, Farmington, USA.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi