Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
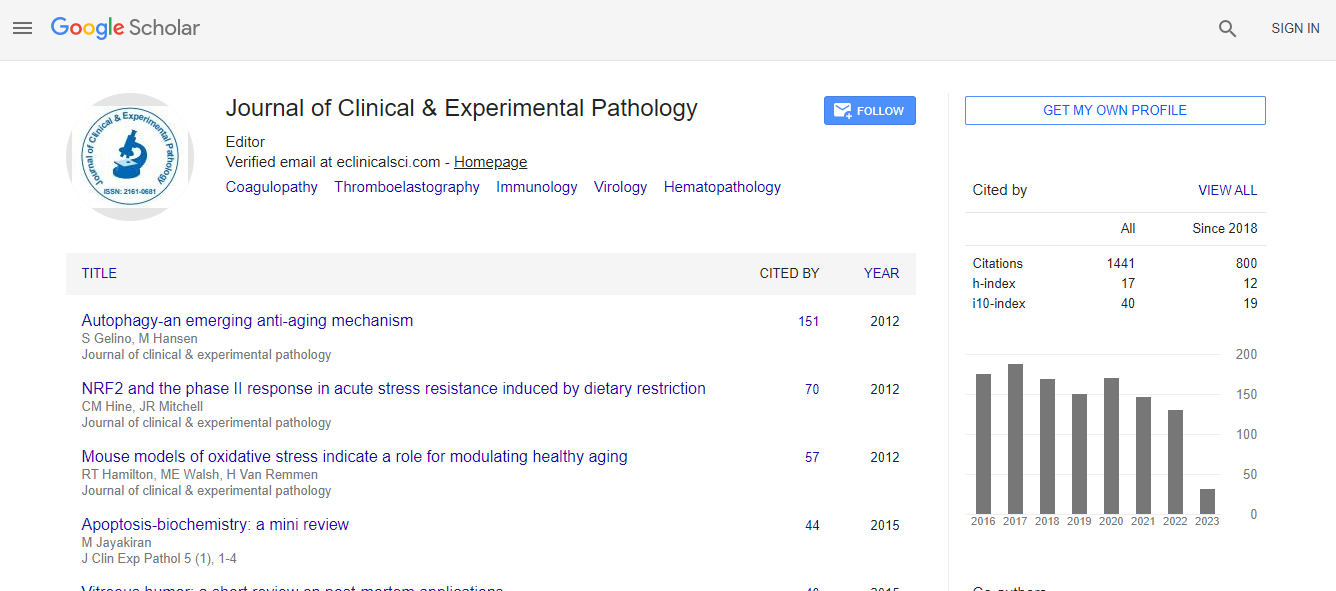
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Failure of redox homeostasis during prion infection
7th World Congress on Molecular Pathology
Cathryn Haigh
The University of Melbourne, Australia
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Prion diseases are invariably fatal neurodegenerative diseases of humans and animals. They are most widely known as a result of their transmissible nature with Creutzfeldt Jakob Disease (CJD) being transmitted from patient to patient through blood transfusion, certain surgical procedures or by use of human-derived hormone therapies and from animals to humans during the bovine spongiform encephalopathy (BSE) outbreak in the UK. The agent that causes these diseases is primary composed of a misfolded protein, the prion protein, which through further templated misfolding events can create more disease-associated forms resulting in disease transmissibility. Whilst the role of the prion protein in disease transmission and progression is firmly established, still very little consensus on the pathways that cause cell death during disease has been reached. Oxidative stress is a feature of prion disease with markers of oxidative damage appearing in the brain in parallel with the detection of mis-folded protein. Data generated by our group has shown how cellular redox homeostasis changes as prion infection progresses from acute to chronic to eventual cell death. The results suggest that prion propagation exacerbates an apoptotic pathway whereby mitochondrial dysfunction follows mislocalization of the critical anti-oxidant enzyme superoxide dismutase-2 (SOD2) to cytosolic caspases, accelerating its degradation. Increased activity of another SOD family member, SOD1, initially compensates for reduction in SOD2 but eventually cellular capacity to maintain oxidative homeostasis is overwhelmed, thus resulting in cell death.Biography
Cathryn Haigh is a Senior Research Fellow at the University of Melbourne, Australia. She was awarded her PhD from the Bath University, UK in 2006 and has been researching in the area of Dementia and Prion Diseases for over ten years. Her research to date has culminated in publication of over 30 papers, more than 20 invited presentations and provision of expert advice for funding agency working groups and research panel discussions.
Email: chaigh@unimelb.edu.au

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi