Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
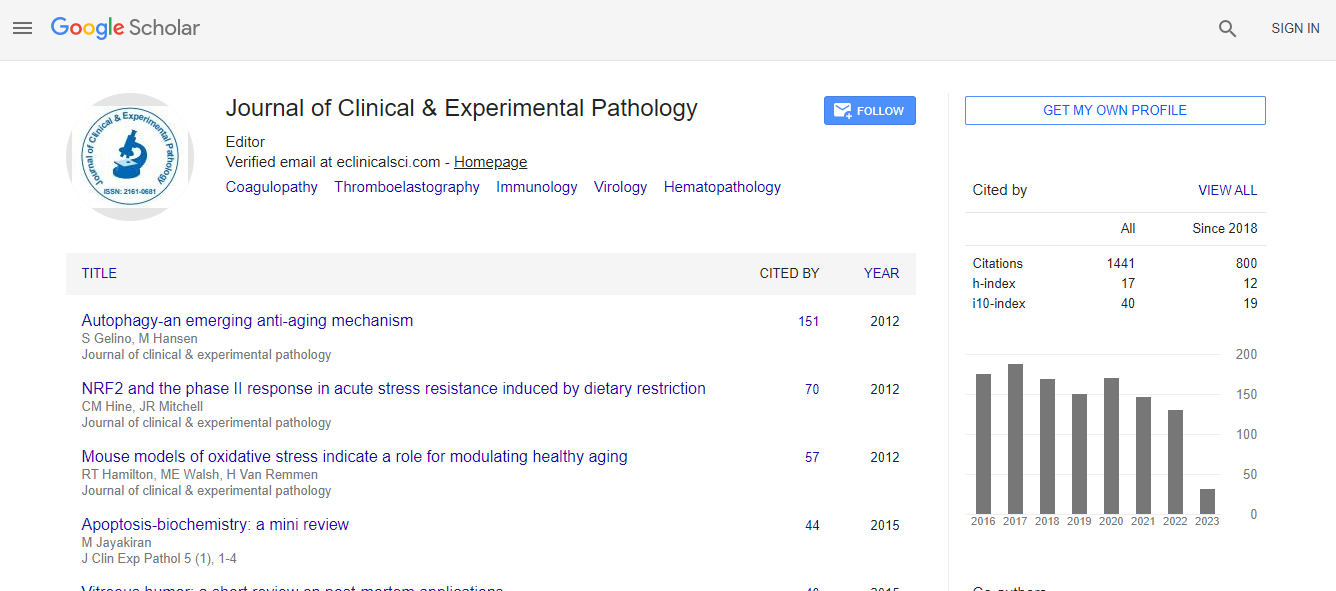
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Genome-wide profiling reveals hyper-trimethylation of histone h3 lysine 27 at PU.1 targets in Myelodysplastic Syndrome (MDS)
International Conference on Pathology
Jason X. Cheng
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
PU.1 is critical for myelopoiesisand histone 3 lysine 27 trimethylation (H3K27me3) isimportant for modulating self-renewal of hematopoietic stem cells (HSCs), but a possible role forPU.1 and H3K27me3 in myelodysplastic syndrome (MDS)has not been illustrated. By focusing on refractory cytopenia with multilineage dysplasia (RCMD) with normal cytogenetics, our epigenetic profiling showed approximately 4600 annotated gene promoters with increased levels of H3K27me3 in RCMD, compared to normal. Computational analysis of the DNA sequences in the hyper-H3K27me3 regions revealed a statistically significant enrichment of the PU.1 binding DNA motif (PU-box). Furthermore, an inverse relationship exists between the hyper- H3K27me3 and PU.1 binding as well as the mRNA expression of PU.1 down-stream myeloid genes at the genome-wide scale. Three PU.1 isoform encoding genes,namelySPI1, SPIB, and SPIC, show different patterns of H3K27me3 at their promoters. By using an erythroid/myeloid line derived from a patient with MDS, we demonstrated that H3K27me3 inhibitors can promote erythroid differentiation. H3K27me3 inhibitors can effectively induce expression of PU.1 and its down-stream genes in both a promyelocytic line and primary MDS bone marrow cells by reducing H3K27me3 at the gene promoters, which strongly suggests epigenetical dysregulation of PU.1 pathway is instrumental in the pathogenesis of MDS.Biography
Dr. Cheng received an M.D. and completed training as a resident surgeon in China. He received a Master in Pathology and served as an instructor at Beijing Medical University BMU. He joined the Ph.D. program and was awarded a Ph.D. degree at University of North Carolina in 1997. He then joined the research program at Memorial Sloan Kettering Cancer Center. Dr. Cheng completed his residency training in Anatomic and Clinical Pathology and his Hematopathology fellowship training at the University of Chicago in 2008 and 2010, respectively. Dr. Cheng became an assistant professor at University of Michigan in 2010.Dr. Cheng was selected as the recipient of the Pathologist-in-Training Award from the Society of Hematopathology and received the Paul E. Strandjord Young Investigator Award from Academy of Clinical Physicians and Scientists in 2008.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi