Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
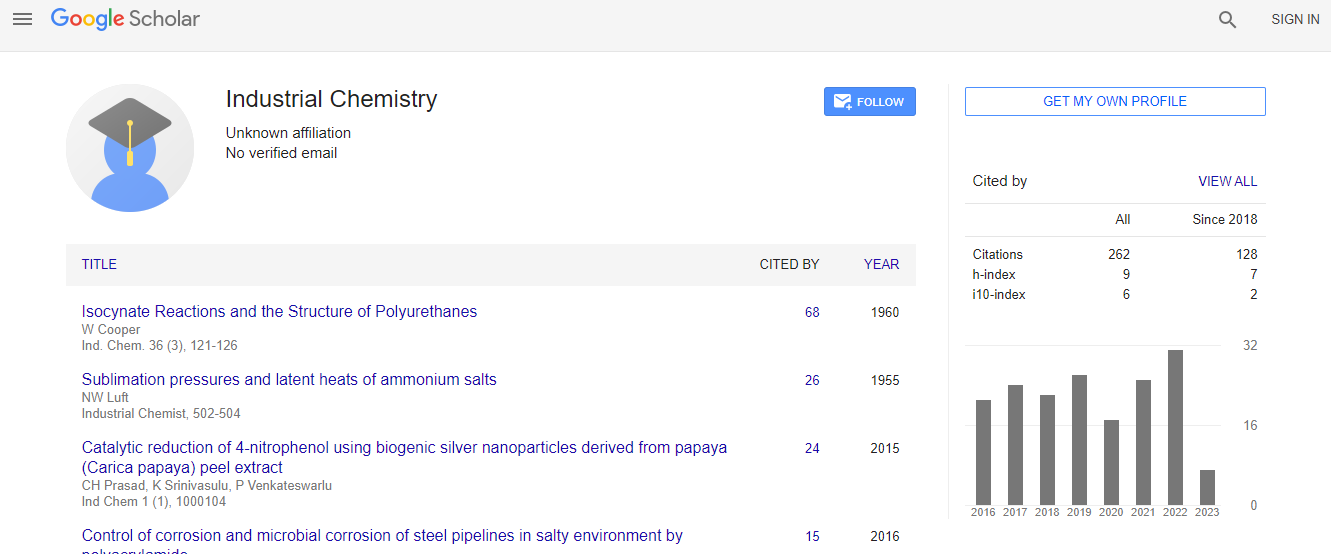
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
High temperature H2S adsorption using copper-titanate nanoparticles
2nd World Conference on Industrial Chemistry and Water Treatment
Farzad Yazdanbakhsh
University of Alberta, Canada
Posters & Accepted Abstracts: Ind Chem
Abstract
Direct desulfurization of syngas is an important measure to further increase the efficiency of IGCC systems. Solid-phase, metal oxide adsorbents which sequester the sulfur by converting H2S to a metal sulfide are the only desulfurization technology capable of withstanding the combustion temperatures present at the outlet of the gasifier. Copper oxide is of particular interest due to its favorable thermodynamics across a wide range of temperatures. Cu-ETS-2 is a copper exchanged form of the sodium titanate ETS-2 and functions analogously to CuO for the conversion of H2S into CuS at temperatures ranging from ambient to 950 �?°C. The results of this study show that Cu-ETS-2 is capable of removing H2S from H2S/He mixture to concentrations below a mass spectrometerâ�?�?s detection limit at temperatures as high as 950 �?°C. Temperature is, however, only one of the challenges facing a direct desulfurization adsorbent; high concentrations of H2 and water vapor are present in the syngas stream which can influence the oxidation state of the metal and the efficiency of H2S removal. In an attempt to prevent reduction of CuO, chromium was successfully used to stabilize the oxidation state of copper oxide and maintain constant adsorption capacity throughout the whole temperature range. While several studies have examined the effect hydrogen in the feed, there are few studies exploring the influence of water vapor on the efficiency of H2S removal and none that explore the effect of water vapor at elevated temperatures. This study can be considered the only study to investigate the influence of water vapor on the desulfurization of a dilute H2S stream at temperatures between 350 and 950 �?°C using copper oxide-based adsorbents. The findings demonstrate that the presence of water vapor promotes production of H2, resulting in faster reduction of CuO to Cu2O and elemental copper, leading to less adsorption capacity. Finally, the ability of the adsorbent for regeneration and use as a multi-cycle adsorbent was investigated. The results indicate that the adsorbent is capable of regeneration for at least four times with no sign of reduction in capacity. The results also indicate that the exothermic nature of oxidation reaction results in temperatures up to ~1700 �?°C causing the partial melting of the quartz glass tube. However the adsorbent can withstand such high temperatures and does not lose adsorption capacity after the first oxidation step. This phenomenon is due to having nano titanate ETS-2 as the support in the adsorbent.Biography
Email: fyazdanb@ualberta.ca

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi