Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
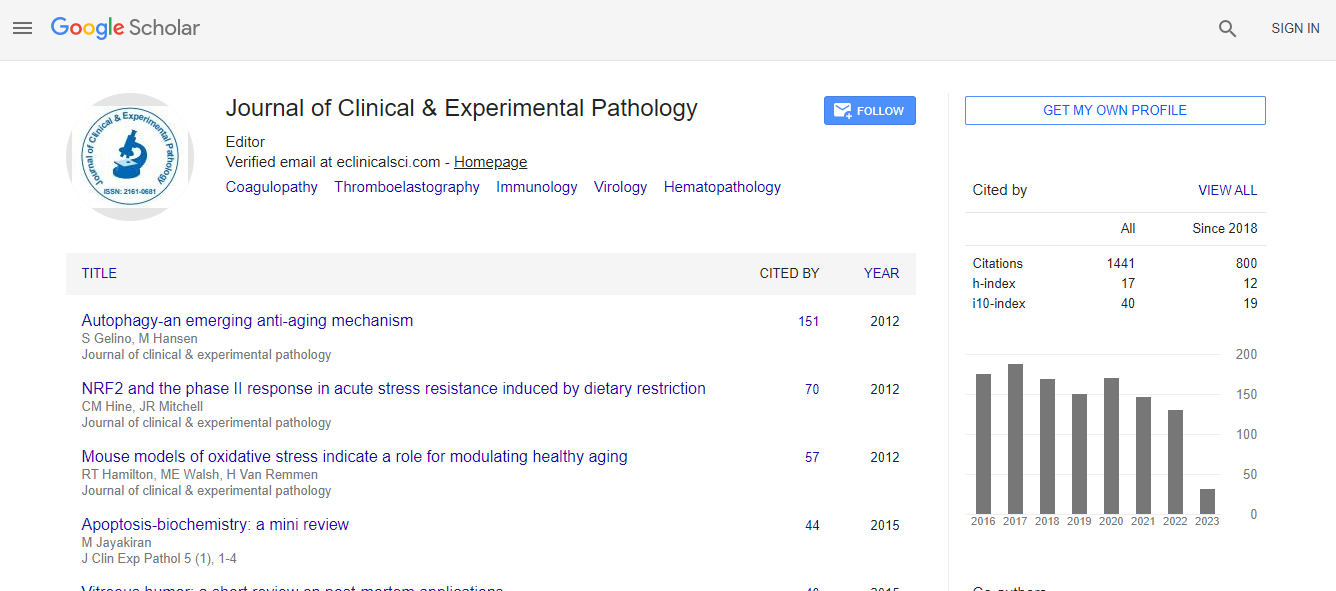
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Mechanisms of the progression from Barretts esophagus to esophageal adenocarcinoma: Role of NADPH oxidase NOX5-S
Joint Event on 15th International Congress on American Pathology and Oncology Research & International Conference on Microbial Genetics and Molecular Microbiology
Weibiao Cao
Rhode Island Hospital and Brown University, USA
Keynote: J Clin Exp Pathol
Abstract
Esophageal adenocarcinoma (EA) has increased in incidence over the past several decades and is characterized by a poor prognosis. Gastroesophageal reflux disease (GERD) complicated by Barrett’s esophagus (BE) is a major risk factor for the development of EA. There is a progression from BE to dysplasia and to EA. However, the mechanisms of progression from BE to EA are not fully understood. We found that NOX5-S is present in Barrett’s cells BAR-T and EA cells FLO and OE33 and is overexpressed in FLO cells and EA tissues. NOX5-S mRNA is also increased in Barrett’s mucosa with high-grade dysplasia. Pulsed acid treatment significantly increases NOX5-S expression and H2O2 production in BAR-T and OE33 cells and Barrett’s mucosa. These data suggest that NOX5-S may be a source of overproduction of reactive oxygen species (ROS) in BE and in EA cells. We also found that acid-induced increase in NOX5-S expression may depend on activation of Rho kinase, ERK1/2 MAP kinases, and cAMP response element-binding protein. The acid may also induce production of platelet-activating factor, which activates signal transducer and activator of transcription 5 (STAT5) and then upregulates NOX5-S. In addition, NOX5-S mediates an acid-induced increase in cell proliferation in Barrett’s cells BAR-T and EA cells (OE33 and FLO). NOX5-S-mediated increase in cell proliferation may depend on the activation of COX2 and microsomal prostaglandin E synthase 1 (mPGES1), and downregulation of p16 via promoter methylation. NOX5-S also mediates acid-induced DNA damage. These data suggest that persistent acid reflux present in BE patients may upregulate NOX5-S, increase production of ROS and cell proliferation, and cause DNA damage, thereby contributing to the progression from BE to dysplasia and to EA.Biography
Weibiao Cao obtained his MD degree from Zhejiang Medical University in Hangzhou, China in 1986. He worked as a Research Associate and an Assistant Professor in Division of Gastroenterology, Rhode Island Hospital, RI, the USA from 1996 to 2007. He was trained as a Pathology Resident from 2007 to 2011 and a GI pathology fellow from 2011 to 2012 in the Department of Pathology, Rhode Island Hospital and Brown University, USA. He has been an attending pathologist since 2012, an Associate Professor since 2013 and a Director of the autopsy service since 2017 in Department of Pathology, Rhode Island Hospital and Brown University, USA. He has been a Member of Oncology C (ONCC) Scientific Peer Review panel, Department of Veterans Affairs, USA and an Editorial Board Member of Scientific Reports (impact factor 4.26) since 2015.
E-mail: weibiao_cao@brown.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi