Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
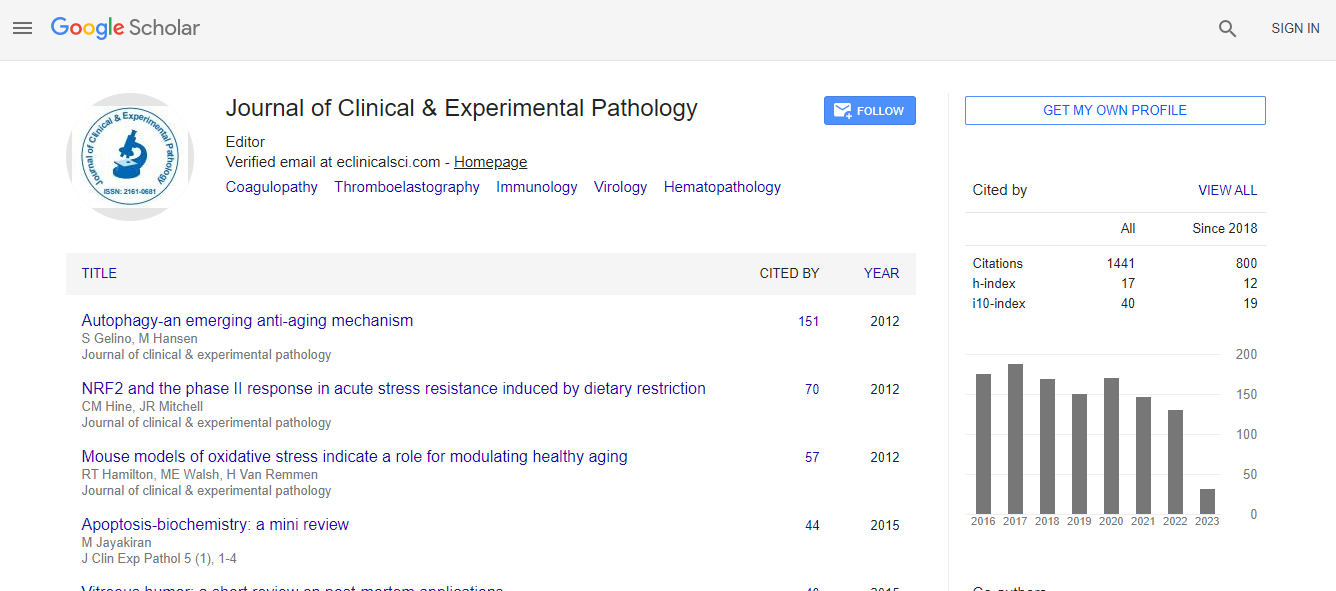
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Molecular tumor boards: An evolving practice of medicine
5th International Conference on Pathology
Rajyasree Emmadi
University of Illinois Hospital & Health Sciences System, USA
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
With the debut of Next Generation Sequencing (NGS) in the CLIA labs, additional specialized information has now become available to the multidisciplinary groups involved in individual patient treatment planning and therapy with regards to oncology. Traditionally, these discussions took place in organ-specific Tumor Boards (such as Breast/GI/GU/Head & Neck, etc.). Many institutions have begun to conduct ��?Molecular��? Tumor Boards that allow for in-depth analysis of NGS data. This data allows unprecedented access to the actual biology of the tumor, permitting design of therapies targeting the achilles heel of each tumor, moving away from the practice of blanket toxic therapies. Discussions are focused on ��?actionable� mutations and specific targeted therapies, possibly through clinical trials. At our institution we have initiated a ��?Precision Medicine��? Tumor Board that goes through this process of NGS data analysis and treatment selection. This lecture will describe the function of the Molecular Tumor Board with actual case examples with a detailed walk-through of the tumor biology and how to match up with newer (clinical trial) treatment options. Some of the limitations that accompany this massive new data source will also be discussed.Biography
Rajyasree Emmadi is a diplomate of the American Board of Pathology in Anatomic & Clinical Pathology as well as in Molecular Genetic Pathology and is an Associate Professor in the department of Pathology at the University of Illinois at Chicago College of Medicine. She is a member of the Professional Relations Committee of the Association for Molecular Pathology and has participated in efforts to preserve the integrity of the practice of Laboratory Medicine.
Email: emmadi@uic.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi