Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
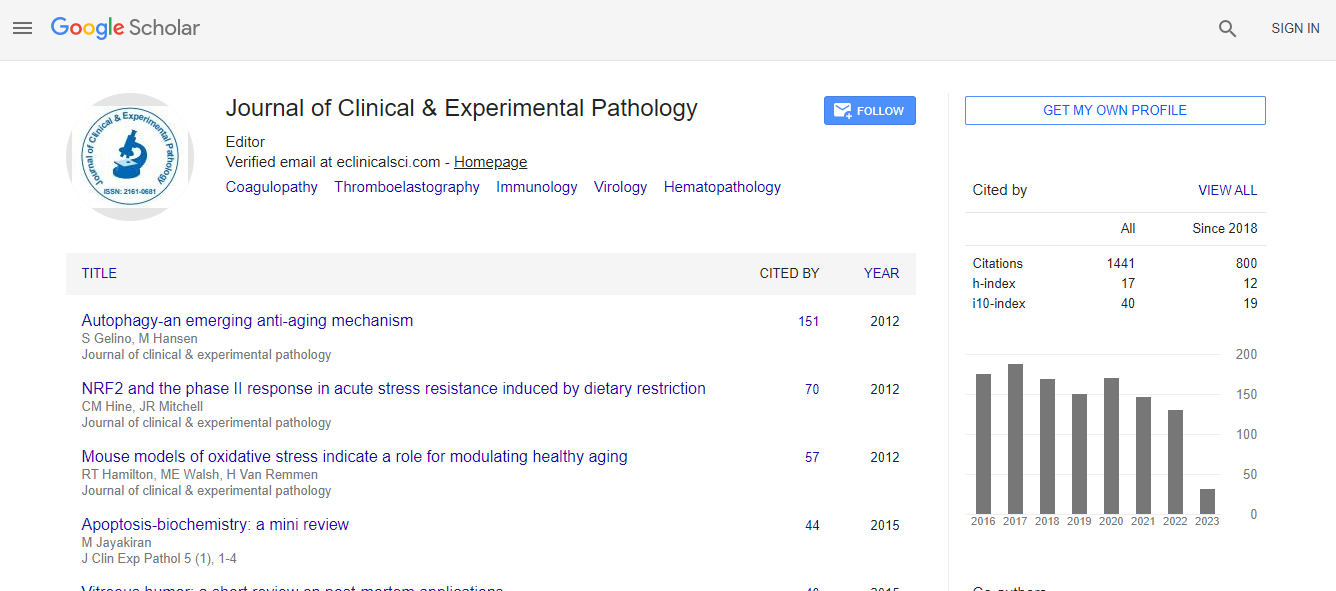
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Peering into the iron window of Alzheimer√¢¬?¬?s disease MR imaging and pathophysiology
13th European Pathology Congress
Jonathan J Wisco
Brigham Young University, USA
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
The severity of pathological protein deposition and concomitant iron presence distinguishes neurological disorders. Tissues with high amounts of protein or iron deposits have a characteristically rapid T2* MRI signal decay. Therefore, these tissue components do not appear on traditional MRI, as the NMR signal has already gone through multiple time constants of decay before any signal can be acquired. Ultra-short Echo Time (UTE) imaging, however, significantly reduces the time between the appearance of an NMR signal and its sampling, allowing for the measurement of iron-related pathology. We used a novel UTE sequence with a 3D cones k-space trajectory in a 3T Siemens scanner to image short T2* tissues in the amygdala and hippocampus in ex vivo, 20 mm thick coronal human brain slabs, each with known Alzheimer√¢¬?¬?s disease (AD) Braak VI tauopathy or with cerebrovascular disease (CVD). We quantified the MR signal from tissues with T2* values of less than 1 ms at TEs of 0.25, 0.5, 0.8, 1.0, 2.0, 3.0, and 5.0 ms and TR of 12.1 ms (1 mm ISO, FA=15 degree, FOV=15 cm2). Different images were then formed by subtracting the TE=5 ms images from the images acquired at the other TEs, effectively suppressing longer T2* tissues. T2* value in the AD amygdala and hippocampus as 4.8+/-1.9 ms (mean+/-SD), and T2* values in anatomically matching regions of the CVD brain was 2.2+/-1.1 ms. We analyzed tissue sections in these regions for the presence of Abeta-42, tau, and CD-68 immunohistochemical reactivity, and enhanced Perl√¢¬?¬?s staining. We noted that the T2* signal decreased with the additive presence of amyloid plaques, tau tangles, non-heme iron, and activated microglia. UTE imaging may be a feasible method to visualize iron-related protein pathology. Future work will further examine the individual contributions of pathological proteins, non-heme iron, and inflammation to the T2* decay.Biography
Jonathan J Wisco is an Associate Professor and Director of the Laboratory for Translational Anatomy of Degenerative Disease and Developmental Disorders, College of Life Sciences, Department of Physiology and Developmental Biology, and Neuroscience Center at Brigham Young University, Provo, UT. He is also an Associate Director of the BYU MRI Research Facility. He holds an Adjunct Associate Professor position in the Department of Neurobiology and Anatomy at University of Utah School of Medicine, USA.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi