Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
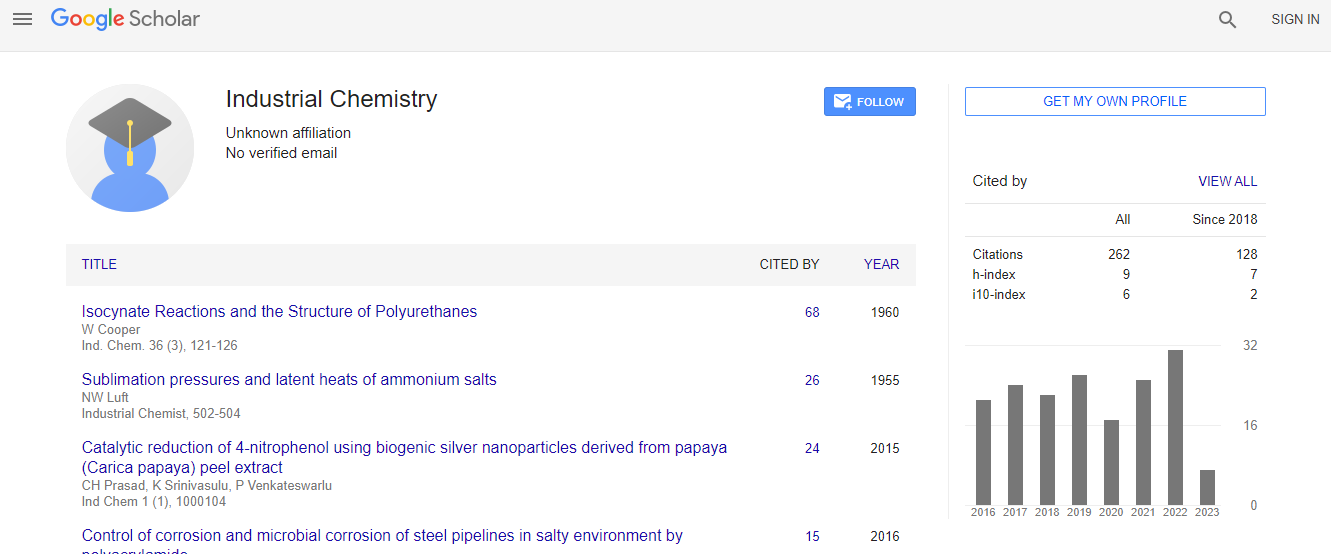
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Precise synthesis of substituted polyacetylenes
2nd World Conference on Industrial Chemistry and Water Treatment
Fumio Sanda
Kansai University, Japan
Keynote: Ind Chem
Abstract
Substituted polyacetylenes have received considerable attention, owing to their properties resulting from the �?-conjugated backbone such as photoconductivity and electroluminescence. The introduction of functional groups at the side chains provides polyacetylenes with useful structural features including liquid crystallinity, molecular recognition, stimuliresponsiveness and gas permeability. Substituted polyacetylenes are synthesized by the polymerization of the corresponding acetylene monomers using transition-metal catalysts. Rh catalysts bearing a triphenylvinyl group polymerize substituted acetylene monomers in a living fashion to give polymers with controlled molecular weights, geometry and end structures. Pd catalysts bearing bulky phosphine ligands polymerize disubstituted acetylene monomers via the coordination-insertion mechanism. Substituted polyacetylenes bearing chiral substituents adopt helical conformations, whose predominant screw sense transforms between right and left-handed in CHCl3/MeOH with respect to the solvent composition. The polymers prefer conformations with large dipole moments in polar media, confirmed by the semi empirical molecular orbital calculations, COSMO method.Biography
Fumio Sanda is a professor in Kansai University. His research studies includes Polymer Synthesis, Transition Metal Catalyzed Polymerization, Conjugated Polymers, Optically Active Polymers. He has received Nakamura award in the year 1997 and Award of Japan Thermosetting Plastics in 2014 and many more.
Email: sanda@kansai-u.ac.jp

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi