Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
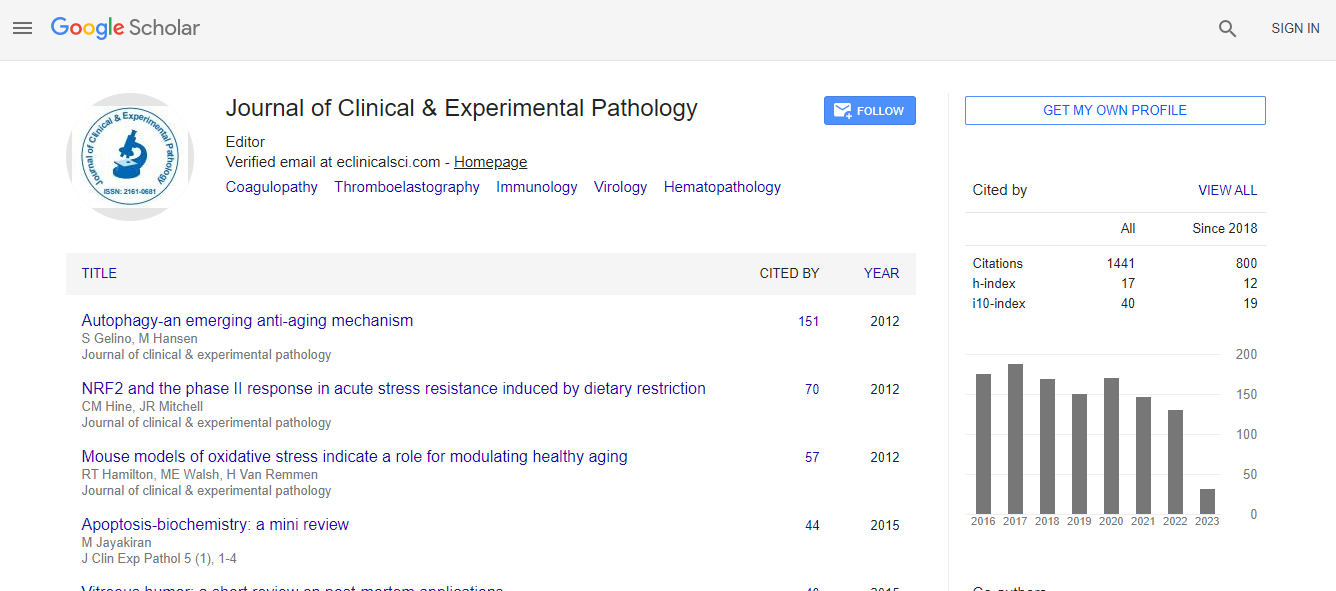
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Quality assurance tools to help reduce the diagnostic interpretive error in breast cancer surgical pathology
Joint Event on 6th World Congress and Expo on Breast Pathology and Cancer Diagnosis & 20th International Conference on Medicinal Chemistry and Rational Drugs
Mark Priebe
Quality Star LLC, USA
Keynote: J Clin Exp Pathol
Abstract
Objective: To review the frequency and related impact of interpretive errors in anatomic pathology and how quality assurance (QA) programs measure in their ability to help reduce diagnostic interpretive error in surgical pathology. Design: From an extensive number of published studies, the rate of major discrepancies identified for cancer patients referred to another institution occur from 4.6% to 14.7%, depending on type of tissue. However published data indicates the current intra-lab QA programs ability to detect these discrepancies is only 0.8% to 1.7%. To help understand the cause of this gap, four formal quality assurance case review programs both inter and intra-lab were reviewed for their ability to satisfy a set of selected design attributes known to help identify interpretive error. Peer reviewed literature was researched to support claims for each program percent compliance to the attributes, strengths, drawbacks, and best demonstrated practices were identified. Results: No program met the selected attribute listing 100%, compliance ranged from 29% (met 2 of 7) to 86% (met 6 of 7) for each program. Conclusion: Laboratories should be aware of the limitations of each QA program and take into consideration their case and pathologist mix and current on-site concerns, select a program with attributes that best match their QA needs. In general, programs that are standardized, include external review by subspecialist and are performed close to the final sign-out date may offer the greatest amount of error discovery and potential to positively influence patient outcomes and continuous improvement. Although this study focused on discordance in cancer related surgical pathology, case review can also be an effective tool in discovery of all histology/cytology diagnostic and clerical discrepancies.Biography
Mark Priebe is a subject matter expert in the utilization of whole slide digital imaging for quality assurance of surgical pathology for cancer. Mark has presented on quality in surgical pathology via podium and posters at multiple scientific meetings and was the Co-Chair for Pathology 16 (Chicago) and Keynote at 5th International Meeting on Breast Pathology and Cancer Diagnosis 2018 (Miami), Mark received his undergraduate degree in Medical Technology from Marquette University, Milwaukee, and advance certification by the ASCP in Immunohematology from the Medical College of Wisconsin, Mark is the co-developer of QualityStar quality consortium of Omaha Nebraska. QualityStar is an external peer review quality assurance program for Surgical Pathology, approved by the American Board of Pathology for Part II (SAM) and IV (QA) MOC and multiple other certification/accreditation agencies. The Mission of QualityStar is to support the reduction of major diagnostic discordance in surgical pathology by 5% (7 to 2%) impacting the lifes of over 80,000 patients annually in North America. mark.
E-mail: priebe@qualitystar.net

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi