Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
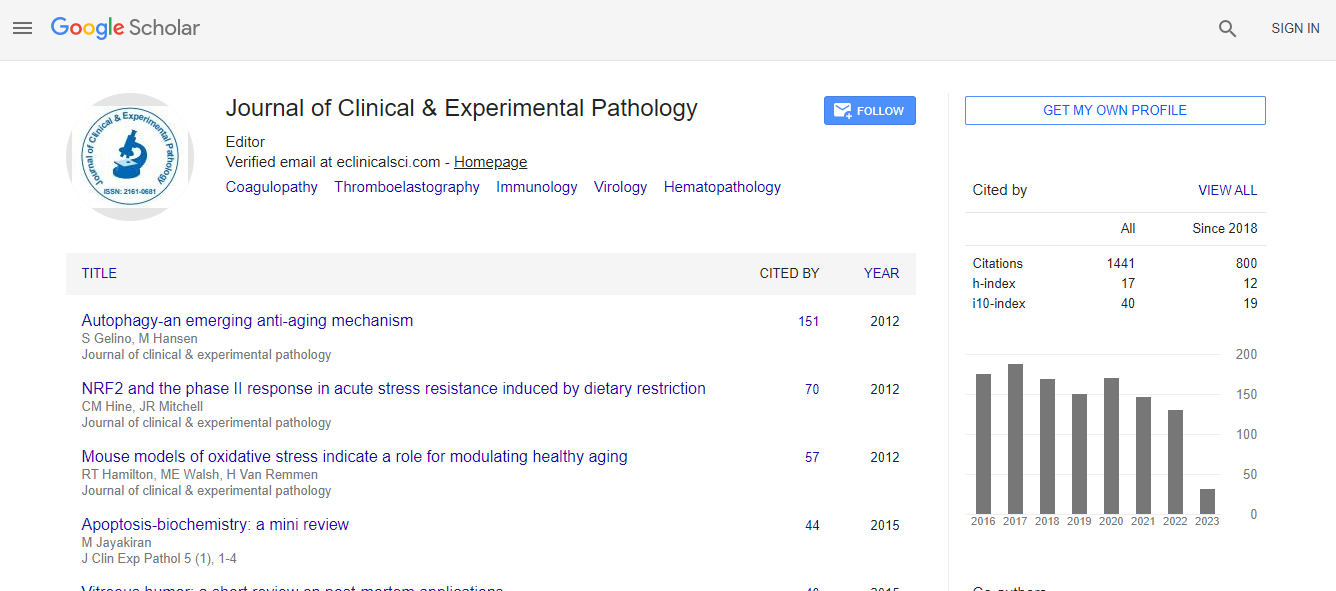
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Regulation of vascular cell function by lipid phosphate phosphatase 3
International Conference on Pathology
Manikandan Panchatcharam
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Meta-analysis of data from a series of genome-wide association studies (GWAS) revealed a striking association between the PPAP2B loci and coronary artery disease. The PPAP2B gene encodes for the lipid phosphate phosphatase 3 (LPP3) integral membrane enzyme, that dephosphorylates lysophosphatidic acid (LPA), sphingosine 1-phosphate (S1P) and related bioactive lipids. While emerging evidence suggests a role for both LPA and S1P in experimental models of atherosclerosis, essentially nothing is understood about the pathophysiologic role of LPP3, largely due to the fact that genetic inactivation results embryonic lethality. We report that constitutive inactivation of LPP3 in vascular cells accomplished by breeding mice with floxed Ppap2b to mice expressing Cre recombinase under control of the Tie1 or SM22 promoter results in dysregulation of LPP3-dependent vascular endothelial cell and smooth muscle cell (SMC) functions which likely underlies the developmental phenotype observed in Ppap2b-null embryos. Using an estrogen inducible Cre transgene under control of the Tie1 promoter, we have inactivated Ppap2b expression in endothelial cells in adult mice. The absence of vascular endothelial LPP3 results in an increase in basal vascular permeability, as assessed by Evans blue dye extravasation in lung tissue. Our observations with respect to SMC suggest that LPP3 serves as an intrinsic negative regulator of SMC phenotypic modulation and inflammation after vascular injury, in part by regulating LPA signaling and act as an important endogenous mediator of development of intimal hyperplasia.Biography
Manikandan Panchatcharam has completed his Ph.D at the age of 31 years from University of Madras and postdoctoral studies from University of North Carolina at Chapel Hill, Carolina Cardiovascular Biology Center. He is an assistant professor at University of Kentucky. He has published more than 23 papers in reputed journals and serving as an editorial board member of repute.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi