Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
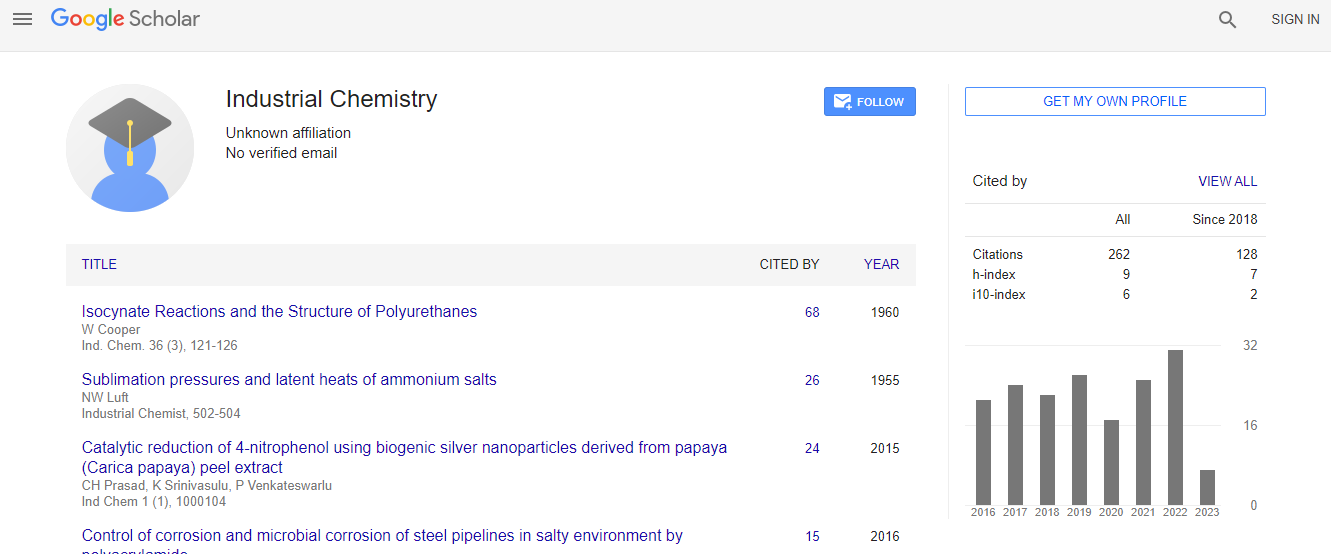
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Synthesis and performance evaluation of Mg/Ce/Mntrimetal oxide-modified diatomaceous earth in groundwater defluoridation
International Conference on Industrial Chemistry
Anthony A Izuagie, Wilson M Gitari and Jabulani R Gumbo
University of Venda, South Africa
Posters & Accepted Abstracts: Ind Chem
Abstract
Groundwater accounts for 25 to 40% of the world��?s drinking water. It is the most preferred alternative source of drinking water for rural communities in the absence of pipe-borne water, being less prone to microbial contamination compared to surface water. However, groundwater as drinking water is known to cause dental and skeletal fluorosis in human if its fluoride content is above the WHO guideline of 1.5 mg/L in drinking water. It is therefore pertinent to reduce the fluoride in groundwater to an acceptable level (0.5-1.5 mg/L) necessary for the development of healthy teeth. In this study, Mg/Ce/Mn oxide-modified diatomaceous earth (DE) was produced by co-precipitation of the metal hydroxides on DE, characterized and applied in batch defluoridation of fluoride spiked water. The sorbent displayed high fluoride removal potentials (> 93% fluoride removal) for solutions with initial fluoride concentration of 10 mg/L - 60 mg/L at sorbent dosage of 0.6 g/100 mL (contact time: 60 min, mixing speed of 200 rpm and temperature: 297 K). The optimum fluoride uptake capacity was 12.63 mg/g for a solution containing initially 100 mg/L fluoride. Percent fluoride removal was above 91% for fluoride solutions with initial concentration of 9 mg/L, over a pH range of ~ 4 to 11, sorbent dosage being 0.6 g/100 mL. The equilibrium pH was greater than the sorbent��?s pHpzc of 5.45. Fluoride removal was therefore by exchange of surface hydroxyl groups by fluoride ions. Sorption data fitted better to Langmuir isotherm and pseudo-second-order model External diffusion was observed to be the sorption rate limiting step.Biography
Email: aaizuagie@gmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi