Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
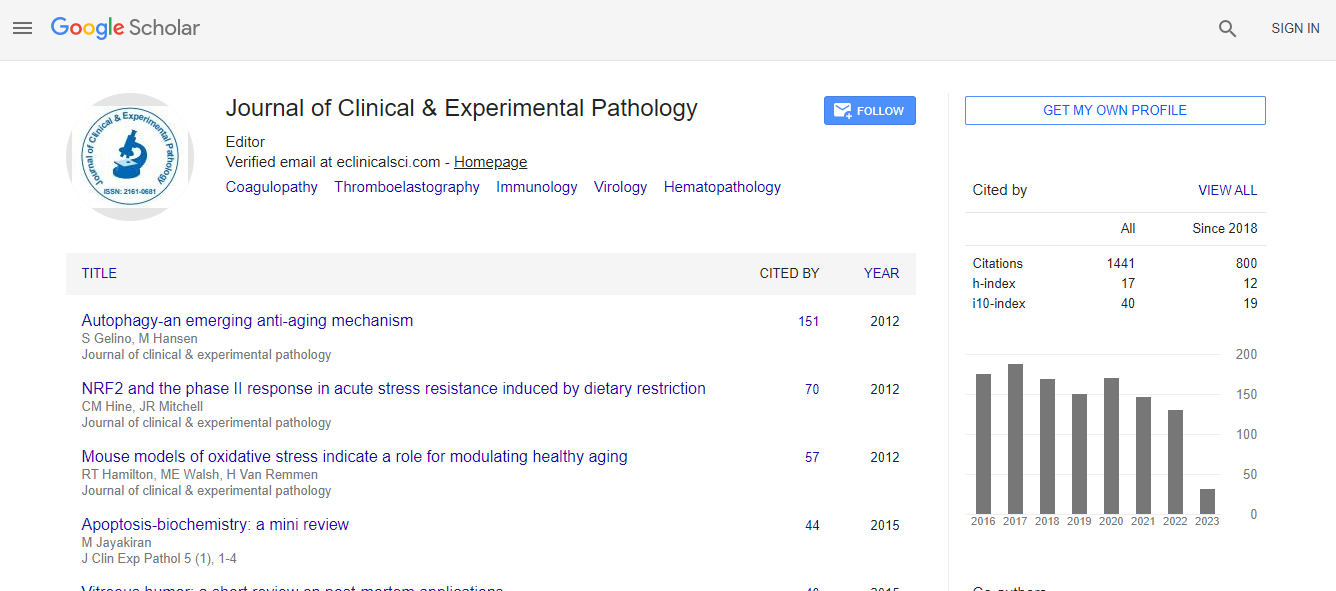
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
The integrated 152 solid tumor panel for early-line and advanced stage treatment decisions in solid tumors
13th International conference on Pathology and Molecular Diagnosis
Melanie Yong
Integrated Molecular Diagnostics Pathology, Inc., USA
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Comprehensive genetic profiling of tumors using next generation sequencing (NGS) is gaining acceptance for guiding treatment decisions in cancer care. We designed a cancer profiling test that integrates results from NGS with more traditional results obtained by immunohistochemistry (IHC) of solid tumor tissues. Relevant regions of genes known to be implicated in solid tumors are targeted for deep sequencing. The tight concordance between some somatic mutations and the standard-ofcare (SOC) therapeutics administered in clinical practice makes identification of such mutations in a specific tumor invaluable in guiding personalized and rational treatment of the patient. The SOC report is provided in a short turnaround time for four tumors, namely lung, breast, colon and melanoma, followed by a full report that includes drug candidates available through clinical trials. For all other tumor types, a full report is provided. Our Integrated 152 solid tumor panel not only detects single nucleotide polymorphisms (SNPs), but will identify copy number variations (CNVs) and some translocations in 152 cancer-related genes. We describe the standardization, validation, and clinical utility of the Integrated 152 Solid tumor test on approximately 250 solid tumor formalin-fixed paraffin-embedded (FFPE) disease samples and control cell-line samples. These studies showed high reproducibility and accuracy (~99%). When therapeutics in clinical trials was included, clinically relevant recommendations increased to 95% for patients in advanced stages of cancer. We present data to demonstrate how the Integrated 152 Solid Tumor Test may be used in clinical practices.Biography
Melanie Yong is the Senior Manager at Integrated Molecular Diagnostics Pathology, Inc. (IMD Path). She earned her Bachelor’s of Science in Biomedical Sciences and Microbiology from Colorado State University and certified by American Society for Clinical Pathology (ASCP) Board of Certification in Molecular Biology, MB (ASCP)CM. She is committed to the highest standard of excellence and enjoys the challenges that come along with new science and technology in the advancing Biotechnology and clinical diagnostics field. Her current research focuses in cancer genomics.
Email: melanie.ypn@gmail.com

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi