Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
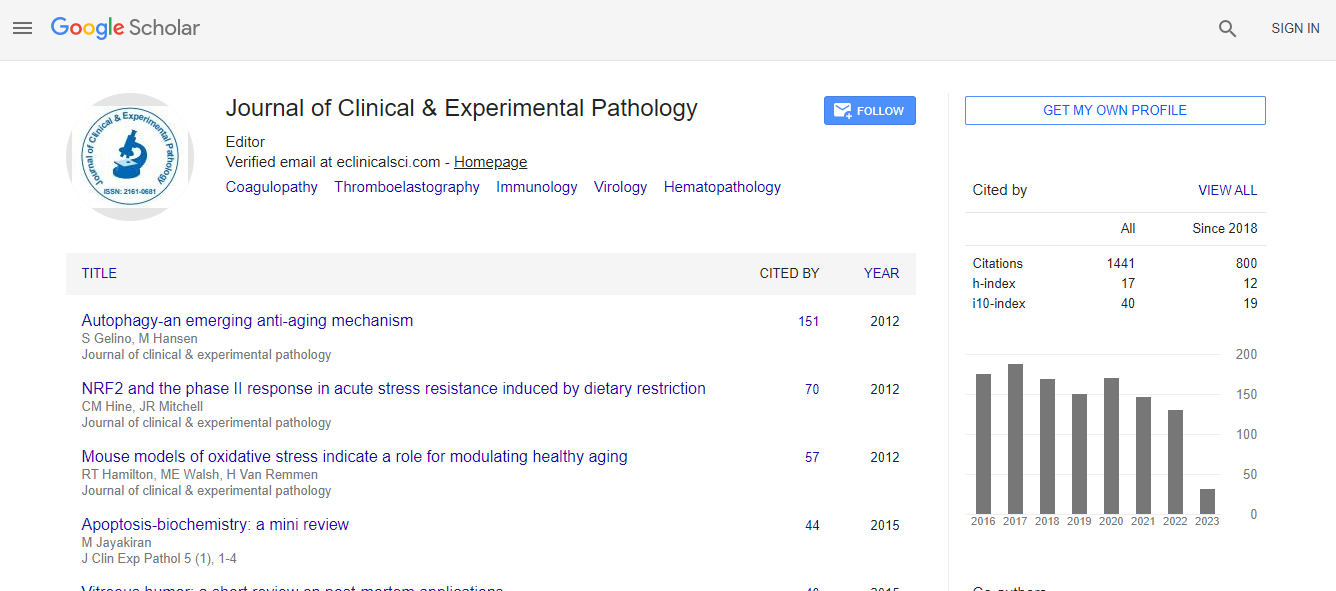
Google Scholar citation report
Citations : 2975
Journal of Clinical & Experimental Pathology received 2975 citations as per Google Scholar report
Journal of Clinical & Experimental Pathology peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Cosmos IF
- Ulrich's Periodicals Directory
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
- world cat
- journal seek genamics
- j-gate
- esji (eurasian scientific journal index)
Useful Links
Recommended Journals
Related Subjects
Share This Page
Understanding the heterogeneity of breast cancer: Referencing the normal breast luminal epithelium
6th European Pathology Congress
Xi Wang
University of Rochester Medical Center, USA
ScientificTracks Abstracts: J Clin Exp Pathol
Abstract
Although molecular sub classification has impacted on breast cancer treatments and outcomes, patient response to targeted therapy or chemotherapy remains highly unpredictable. Dissection of the normal epithelium is fundamental to understanding breast cancer heterogeneity. Terminal ductal lobular unit (TDLU) is the primary site, where the breast carcinogenesis initiates. It consists of two types of cells: Luminal epithelium and myoepithelium. Even inside the luminal epithelium, different cell types are present. For instance, only approximately 10-20% of luminal cells express ER, while the majority of the luminal cells are ER negative. We studied the co-expression of AR and ER in normal breast luminal cells and found that their distribution pattern is the same as what was revealed in invasive breast carcinomas, indicating that ER/AR positive luminal cells may serve as the ��?cell of origin� of ER/ AR positive tumors. These different types of luminal cells could be subjected to different genetic mutations, which could further confound the inter-tumor heterogeneity. We found that Tocopherol-Associated Protein (TAP), a vitamin E binding protein, was co-expressed with ER in normal/benign breast luminal cells, but was down regulated in 46% of ER positive breast carcinomas. This down regulation of TAP was associated with poorer clinical outcome in ER positive breast cancer patients. Further, our study on p53 alteration in breasts of BRCA carriers and non-carriers revealed that p53 positive normal/benign luminal cells were ER negative, indicating that they are the ��?cell of origin� of p53+/ER tumors. We hypothesize that these cells could serve as the ��?p53 signature� to predict future risk of a high grade breast carcinoma.Biography
Xi Wang has completed her Medical education in the Sun Yet-Sen University Medical School in China. She then went to Harvard School of Public Health to be the Research Fellow and later became Research Associate. She has also completed her Residency training in Pathology in West Virginia University and Fellowship training in Sloan- Kettering Cancer Center. She is currently the Associate Professor in Department of Pathology in University of Rochester Medical Center, with the major interests in breast and GYN pathology.
Email: Xi_Wang@URMC.Rochester.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi