Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
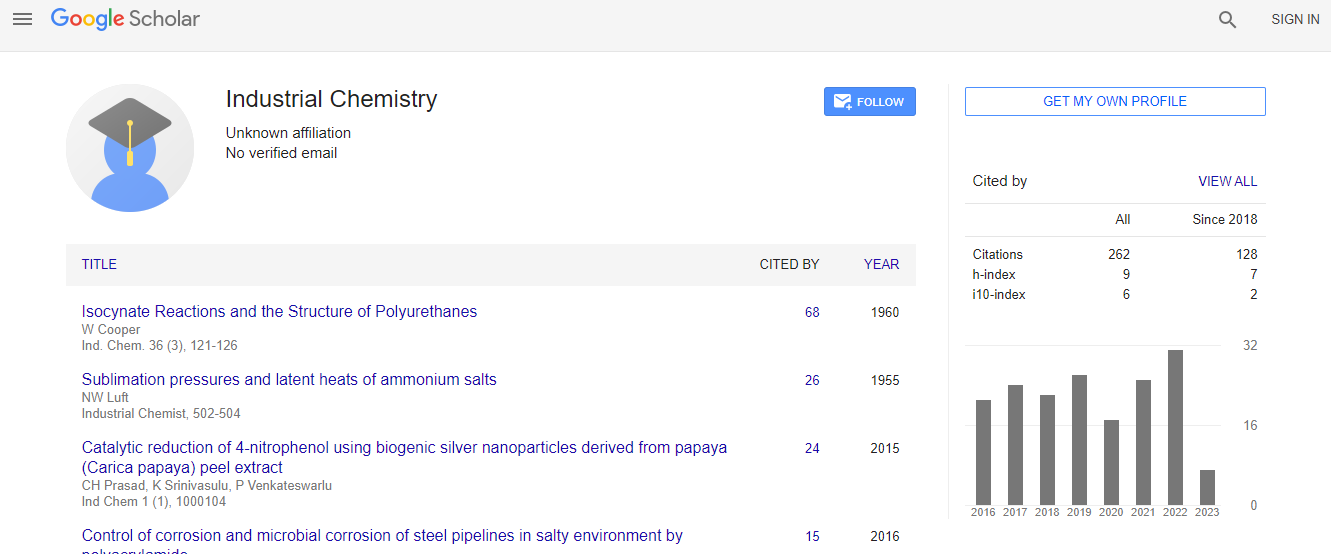
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Unusual phenomena in helix-sense-selective polymerization and its product
2nd World Conference on Industrial Chemistry and Water Treatment
Toshiki Aoki
Niigata University, Japan
Keynote: Ind Chem
Abstract
In 1993, we accidentally discovered the first example of an asymmetric-induced polymerization (AIP) of chiral substituted acetylene, during our study on optical resolution membranes. Since poly substituted acetylenes have no asymmetric carbons, the optical activity is caused by the one-handed helical conformation. Therefore, the optical activity is often not static but dynamic. In 2003, we reported the first helix-sense-selective polymerization (HSSP) of an achiral substituted acetylene by using a chiral catalytic system consisting of chiral phenyl ethylamine (PEA) and a rhodium dimer complex ([Rh (nbd) Cl]2, nbd=2,5-norbornadiene). The achiral monomer had two hydroxymethyl groups and a relatively hydrophobic substituent and the one-handed helicity of the resulting polymer was statically maintained by intramolecular hydrogen bonds in nonpolar solvents. After the discovery, we found some new monomers suitable for the HSSP reaction and have tried to control the polymerization. During these studies, some unusual phenomena were also found to occur in the HSSP process. We found that the HSSP of achiral phenylacetylenes having two hydroxy groups was catalyzed by the chiral polymers prepared by the HSSP of the same or similar achiral phenylacetylenes instead of using the chiral cocatalyst like PEA (Self-HSSP). In addition, it shows the possibility of in-situ self-catalyzed HSSP where the HSSP products formed during the HSSP functioned in-situ as a chiral cocatalyst for the HSSP of the same monomer (In-situ self-HSSP). In addition to the discovery of self-HSSP, other several unusual phenomena were observed in the HSSP of RDHPA. For example, the sense of the helicity of the polymers prepared by HSSP was controlled by changing non-chiral conditions such as the molar ratio of the chiral cocatalyst to the rhodium complex (Reversal HSSP). That may be caused by formation of different chiral rhodium species depending on the ratio. In addition, HSSP was also realized without any chiral source (Spontaneous HSSP). It may result from the formation of a chiral supramolecular compound of the monomer. Since the polymer resulting from HSSP adopts very tight cis-cisoidal conformation and therefore a rigid rod structure, which is not seen in any other poly (substituted acetylenes), it shows many unusual and interesting properties, including unique types of reactivity. For example, in 2013, we reported highly selective photo aromatization (SCAT) of the very tight cis-cisoidal polymer prepared by HSSP.Biography
Toshiki Aoki has received his Doctorate degree (1987) on synthesis and oxygen permeability of graft copolymers from oligosiloxane macromonomers in Department of Applied Chemistry at Nagoya University, Japan. He has joined Fluorine Chemistry Division at Government Industrial Research Institute, Nagoya in 1987-1989. He has then moved to Department of Applied Chemistry at Niigata University in 1989. He has worked at Niigata University as an Assistant Professor (1989), Associate Professor (1995) and Full Professor (2000). He has worked as a Visiting Scientist at University of Southern California from 1998-1999. His research interest covers synthesis of functional polymers, including synthesis of new chiral polymers for permselective membranes. His recent interest is in synthesis and application of two-dimensional polymers using HSSP and SCAT reactions.
Email: toshaoki@eng.niigata-u.ac.jp

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi