Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
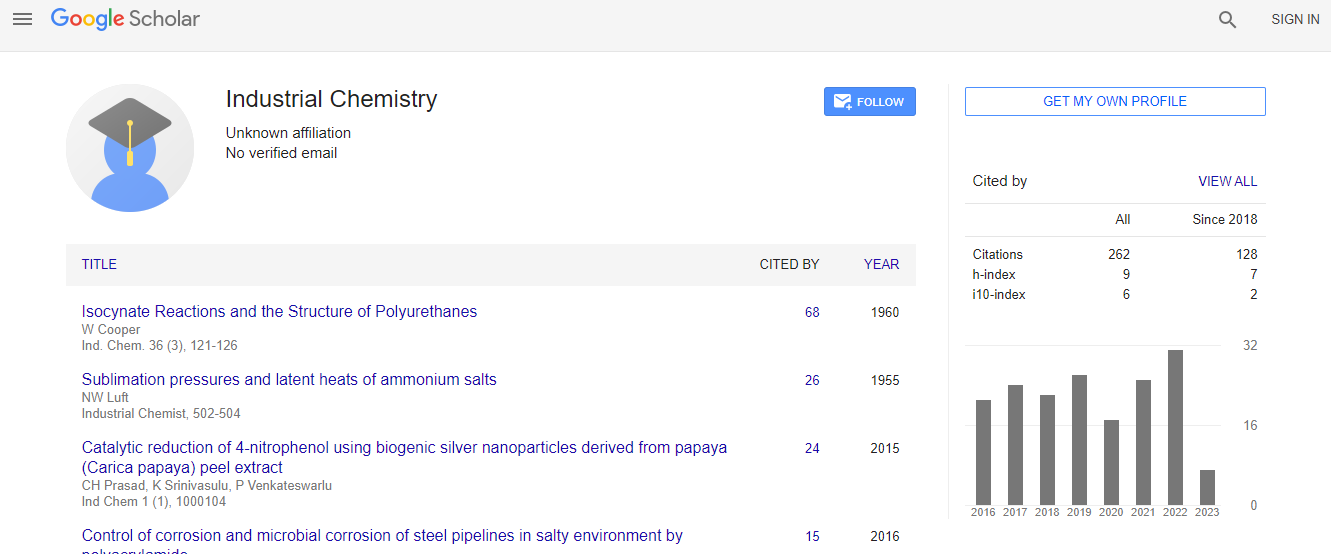
Google Scholar citation report
Citations : 262
Industrial Chemistry received 262 citations as per Google Scholar report
Indexed In
- Index Copernicus
- Google Scholar
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- Geneva Foundation for Medical Education and Research
- Euro Pub
Useful Links
Recommended Journals
Related Subjects
Share This Page
Supermagnetic recyclable Bi/Fe-based nanomaterial with multiple functionalities and its potential practical application in environmental decontamination
International Conference on Industrial Chemistry
Zhong-Ting Hu and Teik-Thye Lim
Nanyang Technological University, Singapore
ScientificTracks Abstracts: Ind Chem
Abstract
Advanced oxidation processes (AOPs) and physical adsorption are efficient and green approaches in environmental decontamination. As everyone knows, TiO2 can drive strong photocatalysis in slurry type and Fe2+ ion can induce Fenton oxidation at pH~3 while there are many investigations on adsorbents (e.g., urchin-like �?±-FeOOH can adsorb 80 mg g-1 of Pb(II), which is much higher than others). However, those used nanomaterials are difficult to separate from the treated water and the post-treatment will be high cost. Herein, we propose a Bi/Fe-based nanomaterial with hierarchical morphology, which can effectively drive AOPs in heterogeneous type at pH~7, has outstanding physical adsorption and supermagnetic property. It can be used to remove organic pollutants and heavy metals, and can be recovered quickly via an environmental-friendly magnetic separation technology. The magnetic property for pristine bismuth ferrites is too weak to be used in practical application effectively. Here Bi/Fe-based materials with coral-like hierarchical morphology were fabricated using solvothermal treatment in methanol system. Its saturation magnetization (Ms) marvelously increase from 0.375 to 30.7 emu g-1, while the adsorption of methyl orange (a dye pollutant) ranges from 0.5 to 46.6%. Besides, it also can effectively induce visible light photo-Fenton oxidation which can be used to degrade different types of organic pollutants (e.g., dyes, pharmaceuticals, pesticides). Even at a low catalyst loading of 0.12 g L-1, the removal rate of organic pollutants can be ~99% in 100 min by degradation and/or adsorption. Its adsorption ability also can be used to remove different kinds of heavy metal ions (e.g., Pb(II), Cd(II), As(V), Cr(VI), Cu(II), Mn(II), Ba(II) and Co(II)), especially for Pb(II), for which its maximal adsorption capacity can reach a new height of 214.5 mg g-1. The outstanding performances are possibly ascribed to its coral-like hierarchical morphology which was investigated by several characterization techniques. It was proved that it is self-assembled by 1D nanowires (~6 nm in diameter) and 2D ultrathin nanoflakes (~4.5 nm in thickness). This product has remarkable optical properties with absorption of UV, visible light and even IR as well.Biography
Zhong-Ting Hu is a PhD student from Nanyang Technological University (Singapore). He holds BSc in Applied Chemistry and MSc in Environmental Engineering. He was a R&D researcher of NanoMaterials Technology Pte Ltd. in Singapore (2007-2012). He has experiences in nanoparticle synthesis, surface modification, wet coating and nanomaterial production in pilot plant. He was a Team Leader of a research project for undergraduate students (chemical plating & H2 energy) and their paper won the 1st Prize of the 1st ZJNU Natural Science’s Academic Paper Competition. His current research interests are material and environment including advanced nanomaterials fabrication/optimization (morphology, self-assembly, nanocomposite, doping, synthesis), environmental photochemistry, heterogeneous catalysis, water treatment, solar energy, magnetic separation.
Email: zhu009@e.ntu.edu.sg

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi