Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
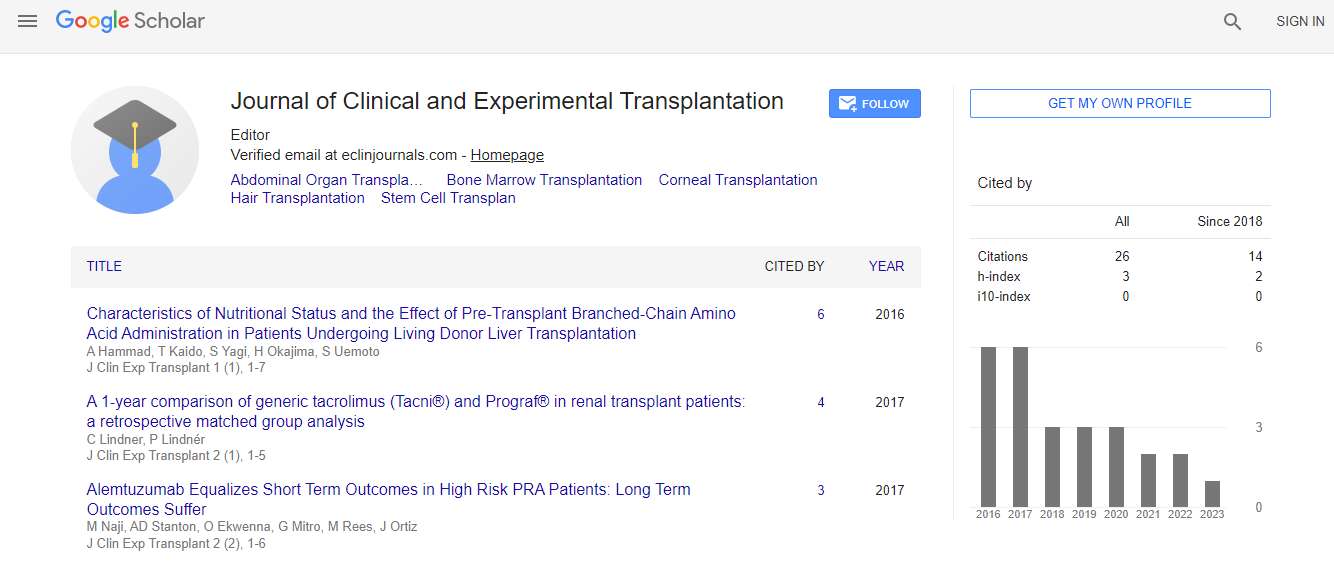
Google Scholar citation report
Citations : 26
Journal of Clinical and Experimental Transplantation peer review process verified at publons
Indexed In
- Google Scholar
- RefSeek
- Hamdard University
- EBSCO A-Z
- Publons
- Euro Pub
- Geneva Foundation for Medical Education and Research
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
James M. Mathew

James M. Mathew
Departments of Surgery and Microbiology-Immunology
Northwestern University Feinberg School of Medicine
Chicago, USA
Biography
James M. Mathew, PhD. is an Associate Professor in the Departments of Surgery and Microbiology-Immunology, and the Director of Immune Monitoring Core at the Comprehensive Transplant Center, Northwestern University Feinberg School of Medicine, Chicago. His research for the past 27 years has been in the areas of immune responsiveness and tolerance in clinical organ transplantation. These studies have resulted in more than 230 peer reviewed publications. The major thrust of these has been on the immunological effects of donor bone marrow cell and donor hematopoietic stem cell infusions in organ transplant patients using in vitro and ex vivo culture systems. One specific observation was that the use of an array of assays for donor specific unresponsiveness could possibly identify potentially tolerant recipients who may be candidates for immunosuppressive drug withdrawal. Recent studies have also assessed the role of CD4+CD127-CD25HighFoxP3+ regulatory T cells as potential tolerance inducers in kidney and liver transplant patients, as well as the differential susceptibility these cells to clinically used immunosuppressive drugs. More recently, he has developed the technology to expand antigen-specific regulatory T cells with a view to using them clinically to induce donor specific tolerance.
Research Interest
Clinical Trial Methodology, Immune Regulation, Immunology; Infectious Diseases-viral, Liver disease, pathobiology, Stem Cells, Translational Research, Transplantation

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi