Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
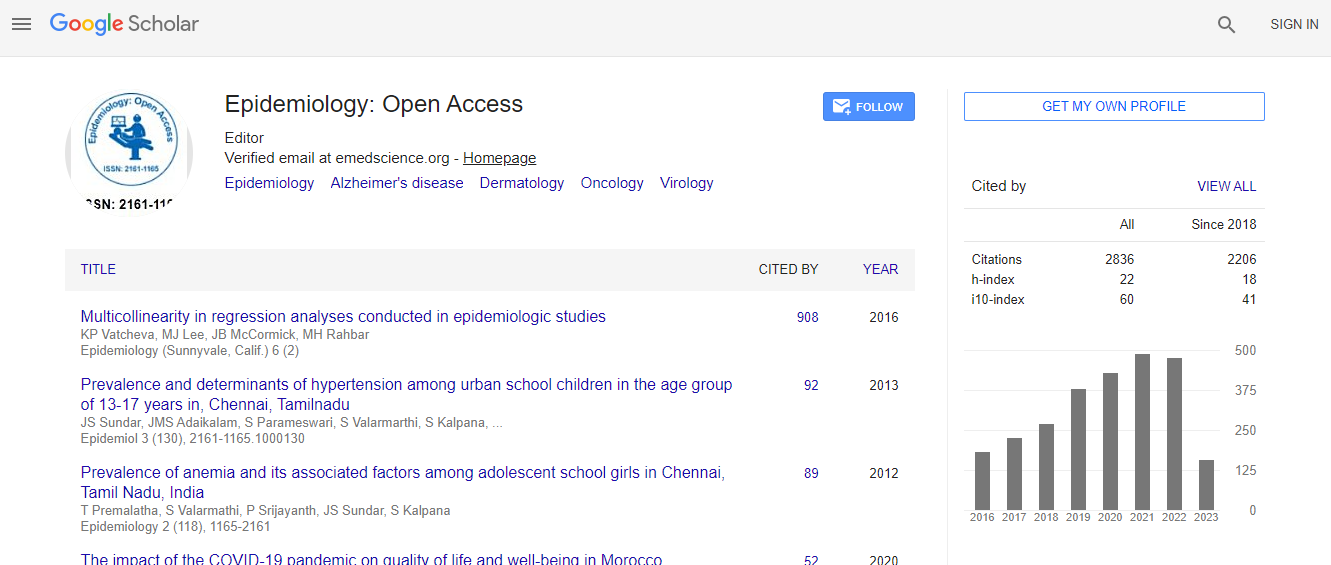
Google Scholar citation report
Citations : 3864
Epidemiology: Open Access received 3864 citations as per Google Scholar report
Epidemiology: Open Access peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Genamics JournalSeek
- SafetyLit
- Access to Global Online Research in Agriculture (AGORA)
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Evaluation of a CFP32-based serological test for the diagnosis of tuberculosis in populations with different levels of exposure
3rd International Conference on Epidemiology & Public Health
Jihene bettaieb, Chaouki Benabdessalem, Dahmani M Fathallah and Ridha M Barbouche
Posters-Accepted Abstracts: Epidemiology (Sunnyvale)
Abstract
Introduction: In previous study we have reported the development of a serological test using Pichia pastoris recombinant
CFP32 antigen, a virulence factor of Mycobacterium tuberculosis (MTB), with both high sensitivity and specificity in the
Tunisian population. In this study, we aimed to assess the ability of the CFP32 serological test, to discriminate between TB
patients and non-TB among people of different ethnic origins.
Materials & Methods: Serum samples were obtained from 224 donors from different countries, divided into two groups: i.
samples from Tunisian cohort (intermediate endemicity) including TB patients and their contacts ii. Samples from WHO
banked sera (obtained from sub-Saharan African and Latin American countries with high endemicity) including TB patients
and TB suspected individuals. Receiver operating curve (ROC) was used to evaluate the performance of the CFP32 serological
test.
Results: CFP32 specific antibody levels in the TB patients was significantly higher than non-TB controls in Tunisian group
(p<10-4), Sub-Saharan African group (p<10-3) and Latin American group (p<10-4). Considering all groups together, the
area of the ROC curve was 0.77 (95%CI, 0.70-0.83). The cutoff level was set at 0.63, giving the optimal combination between
sensitivity (70%, CI95%: 60-79) and specificity (73%, CI95%: 63-81).
Conclusion: Although CFP32 single antigen serological test did not achieve excellent sensitivity and specificity, a combination
of CFP32 with other select antigens and particularly stage-specific infection antigens could be used to improve performance of
a serological test regardless of TB incidence and latent infection.
Biography
Jihene bettaieb is working in Laboratory of Medical Epidemiology, Pasteur Institute of Tunis, Tunisia.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi