Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
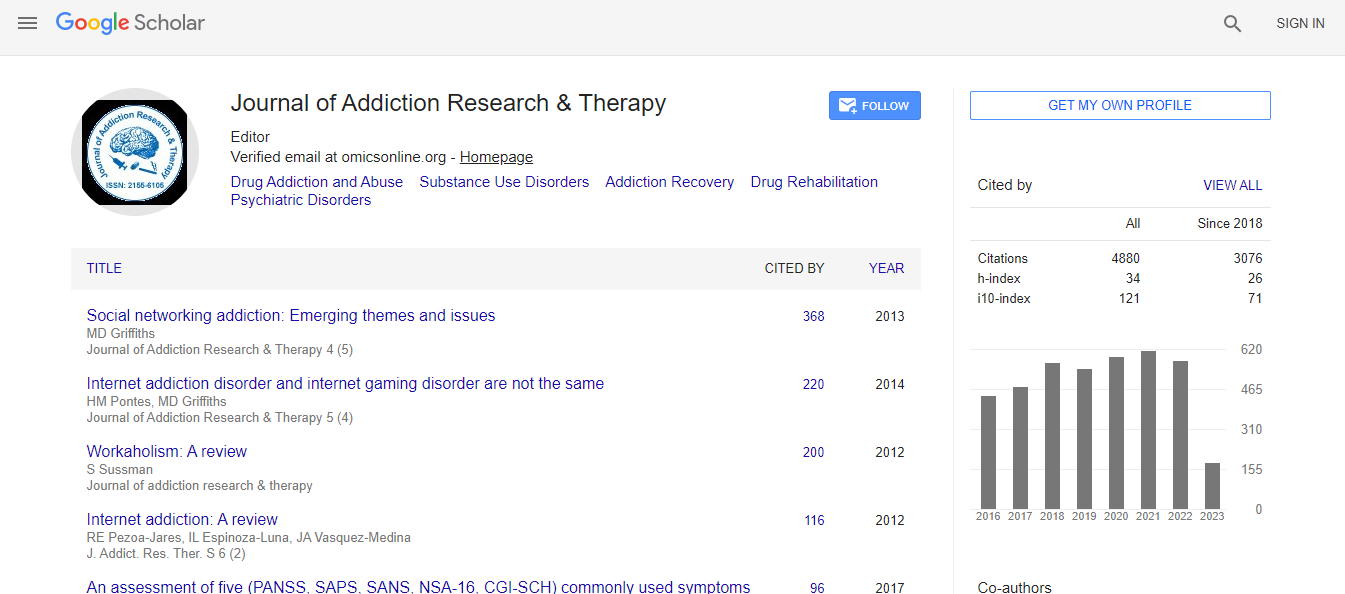
Google Scholar citation report
Citations : 4859
Journal of Addiction Research & Therapy received 4859 citations as per Google Scholar report
Journal of Addiction Research & Therapy peer review process verified at publons
Indexed In
- CAS Source Index (CASSI)
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- SafetyLit
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Elucidating the role of reward mechanisms in pathophysiology and treatment of obesity
4th International Conference and Exhibition on Addiction Research & Therapy
Igor Elman
Keynote: J Addict Res Ther
Abstract
Background: Obesity and overeating may be construed as a behavioral addiction. In schizophrenia, obesity is twice as prevalent as in the general public afflicting over 50% of the patients and shortening their lifespan by about 15 years. Although excessive consumption of fast food and pharmacotherapy with second-generation antipsychotic agents (SGAs) has been implicated in the schizophrenia/obesity comorbidity, the pathophysiology of this link remains unclear. The mechanism proposed here is based on the central opioidergic system owing to opioid��?s role in: a) Enhancing reward features of food; b) Boosting orexigenic and suppressing anorexigenic neuropeptides; c) Reducing peripheral insulin secretion and d) Desensitizing insulin receptors. The purpose of this presentation is to discuss a heuristic value of opioid blockade for patient��?s metabolic status. Methods: Translational evidence to be presented in support of the above contention includes a preclinical and two clinical studies. First, four groups of Wistar Han IGS rats were treated for 28 days with an SGA olanzapine, a combination of olanzapine and an opioid receptor antagonist, naltrexone, naltrexone alone or vehicle, and their food consumption and body weight were measured daily for the first nine days and every other day thereafter. Second, a potential mechanism of naltrexone action was explored in 15 patients with heroin dependence who underwent the standard sweet taste test before and 7 days after the injection of depot naltrexone. Third, we also conducted a double-blind placebo-controlled pilot clinical trial where schizophrenic or schizoaffective patients on a stable dose of olanzapine were randomized in a double-blind fashion to receive naltrexone (n=14) or placebo (n=16). Results: Rats treated with olanzapine and naltrexone were similar to the vehicle-treated animals with respect to food intake and body weight gain, whereas olanzapine treatment alone induced overeating and obesity (p<0.001 group-by-time interaction). Data from heroine dependent human subjects demonstrated a reduction in the hedonic and motivational ratings of sweet solutions (p<0.05) after naltrexone suggesting a potential mechanism of action. On the clinical trial, in comparison to the olanzapine and placebo combination, the olanzapine and naltrexone group displayed a significant decrease in the fat mass (p<0.01), assessed with the biometric impedance analysis and a trend towards improvement in the insulin resistance quantified via HOMA-IR values (p=0.09). Conclusions: Naltrexone addition may result in clinically meaningful attenuation of olanzapine-induced metabolic side effects. Potential mechanisms of naltrexone action may involve diminution of rewarding features of food in conjunction with favorable effects on insulin sensitivity. If confirmed, these results may contribute to the identification of an inexpensive and effective treatment that specifically targets the underlying pathophysiologic effects of SGAs and provides a substantial clinical benefit to the at risk population.Biography
Igor Elman, MD is an Associate Professor of Psychiatry at Harvard Medical School. He was trained at the National Institute of Mental Health and is a Diplomate of the American Board of Psychiatry with a subspecialty certification in Addiction Psychiatry. Presently, he heads a clinical research lab, focused on the role of reward and motivational systems in pathophysiology of severe neuropsychiatric disorders, including addictions, schizophrenia and post-traumatic stress disorder. He is a past recipient of NIDA K23 and R01 Awards. He serves as Editor-in-Chief of the Journal of Psychology Research and Behavioral Management.

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi