Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
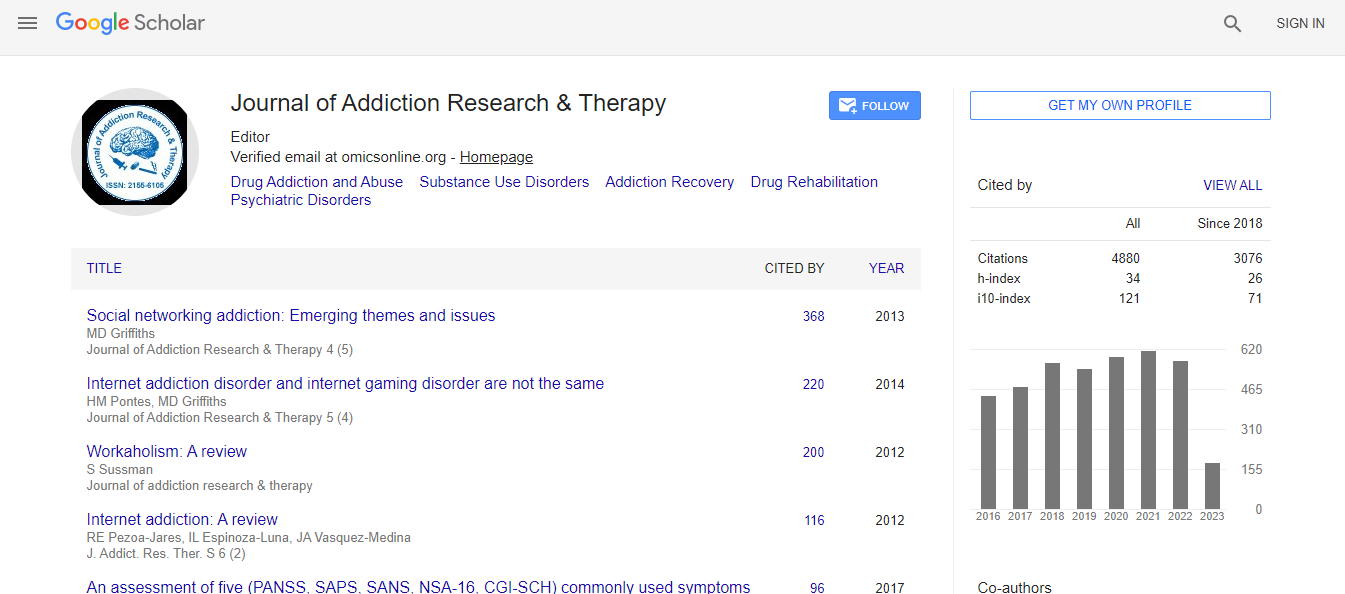
Google Scholar citation report
Citations : 4859
Journal of Addiction Research & Therapy received 4859 citations as per Google Scholar report
Journal of Addiction Research & Therapy peer review process verified at publons
Indexed In
- CAS Source Index (CASSI)
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- SafetyLit
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Programmable bio-nano-chips for quantitation of drugs of abuse in oral fluids
International Conference and Exhibition on Dual Diagnosis
J T McDevitt
Rice University, USA
Keynote: J Addict Res Ther
Abstract
Use of illicit drugs and abuse of licit counterparts are associated with multiple physical health, emotional, and interpersonal problems. Cardiovascular disease, stroke, cancer, HIV/AIDS, anxiety, depression, and sleep problems, financial difficulties and legal complications, work, and family problems can all result from or be exacerbated by drug abuse. In 2013, 21.6 million Americans were dependent on drugs or were drug abusers, representing 8.2% of our population aged 12 and over. In the US each year drug abuse and drug addiction cost employers over $122 billion in lost productivity time and another $15 billion in health insurance costs. Routine screening for substance use disorders could alter this statistic and get more people the help they need. Indeed, since the inception of medication-assisted treatment (MAT) for opioid addiction, drug testing has provided both an objective measure of treatment efficacy and a tool to monitor patient progress. Likewise, increasing emphasis on treatment outcomes as evidence of program effectiveness has added significance to drug tests in Treatment Programs (TPs), as well as use of drug test results in response to quality assurance requirements. Up to this juncture, drug testing in treatment settings has been limited to either remote drug testing via laboratory-confined procedures that take a long time to return results, or on-site urine testing strips that have limited multiplexing and semi-quantitative measurement capacities. There is, thus, a compelling need for an advanced point of care drug testing solution to be implemented within addiction treatment settings. Drug test results help policymakers and TP administrators detect and monitor emerging trends in substance abuse that may signal a need to redirect resources. Drug use patterns have changed markedly in recent decades; for example, benzodiazepines, amphetamines, methamphetamine and cocaine have increased in popularity while barbiturate use has diminished. New substances of abuse or combinations of substances and methods of ingestion present new treatment challenges and funding concerns. This keynote lecture will summarize recent developments related to the development, testing and validation of the Programmable Bio- Nano-Chip (p-BNCs) platform technology and its use in the measurement of drugs of abuse in oral fluids. This powerful new p-BNC technology is used to quantify soluble targets in complex biological matrices, using agarose bead sensors with a sizetunable network of nanometer-scale fibers, arrayed within an enclosed microchip. Unlike existing portable and lab-based drug detection systems, this approach allows for simultaneous detection and quantitation of multiple drugs in ~10 minutes. These mini test ensembles have been shown to exhibit outstanding analytical performance and are now clinically validated against the reference method of Liquid Chromatography Tandem Mass Spectroscopy. The new chip-based tests allowed for the elucidation of the time-course of drugs in oral fluids: cocaine/benzoylecgonine, were measured (LODpractical and LOQpractical of 1.0 and10.0ng/mL, respectively) as early as 10 min and up to ~50 hours post IV infusion, while methamphetamine (LOD=6.2ng/ mL, LOQ=8.1ng/mL) demonstrated clearing 4 hours post its administration. This methodology may find utility in numerous settings including road-side drug testing, drug rehabilitation centers, emergency rooms, prisons, schools, and within the work place.Biography
J T McDevitt serves as the Chair for the Department Biomaterials and Biomimetic at New York University College of Dentistry and is a pioneer in the development of ‘programmable bio-Nano-chip’ technologies. He has a strong track record of translating essential bioscience discoveries into real-world clinical practice. In this capacity, he serves as the Scientific Founder for several companies in areas related to medical micro device technologies. His most recent company, SensoDx, features a universal platform sensor technology with capacity to digitize biological signatures for a broad range of key health conditions. He and his team over the past decade have risen over $25M in Federal and Foundation support. His recent research has been sponsored by major programs funded by the National Institute of Dental and Craniofacial Research (NIDCR) division of the National Institutes of Health (NIH), the Bill and Melinda Gates Foundation, Cancer Prevention Research Institute of Texas (CPRIT), the National Aeronautics and Space Administration (NASA) and the United Kingdom’s Home Office Scientific Development Branch.
Email: mcdevitt@nyu.edu rg12@bcm.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi