Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
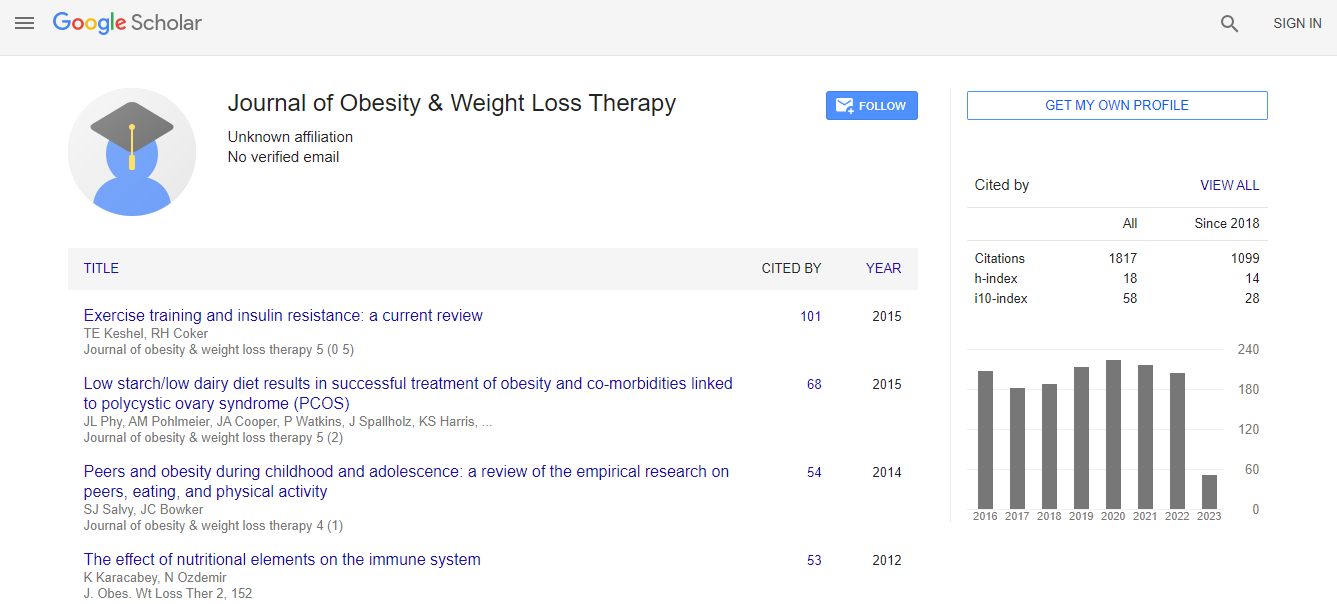
Google Scholar citation report
Citations : 2305
Journal of Obesity & Weight Loss Therapy received 2305 citations as per Google Scholar report
Journal of Obesity & Weight Loss Therapy peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Open J Gate
- Genamics JournalSeek
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- University of Bristol
- Pubmed
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Calmodulin dependent protein kinase (CaMK)II activation by exercise regulates NRF-1 and its target lipid oxidizing target gene, Cpt-1 in rat skeletal muscle
10th International Conference and Exhibition on Obesity & Weight Management
Emmanuel Mukwevho
North West University, South Africa
Keynote: J Obes Weight Loss Ther
Abstract
Regular exercise increases oxidation of fatty acids in skeletal muscle. Exercise activates Calmodulin-dependent protein kinase (CaMK)II, resulting in increased mitochondrial oxidative capacity. As such, exercise can curb accumulation of excess lipids in adipose and intramuscular tissues that may result in obesity/type 2 diabetes. Lipid metabolism mainly occurs in mitochondria regulated by NRF-1 and is controlled by a set of mitochondrial enzymes. For example, Carnitine palmitoyltransferase (CPT)-1 is a rate-limiting enzyme in mitochondrial lipid oxidation that regulates the transport of long chain fatty acids across the mitochondrial membrane, resulting in ATP synthesis. On the other hand, acetyl-CoA carboxylase (ACC)-1 is a mitochondrial enzyme that promotes lipid synthesis by providing malonyl CoA substrate for the biosynthesis of fatty acids. NRF-1 is the major transcriptional factor of the mitochondria, the site for ATP generation from carbohydrates and lipids. As such, mitochondrial dysfunction is crucial in metabolism of the cell. In order to investigate the amount of NRF-1 bound Cpt-1, ChIP assay performed. Exercise showed that the amount of NRF-1 bound Cpt-1 was ~1.3 fold increase compared with the control group. The exercise + KN93 group did not show any significant change compared with the exercise group. This result indicates that exercise-induced CaMKII activation increase the amount of NRF-1 bound Cpt-1. With respective to gene transcription, exercise group showed ~7.8 fold increase compared with the control group. Cpt-1 gene expression of the exercise + KN93 group showed significant decrease compared with the exercise group. Cpt-1 gene expression of the exercise + KN93 was similar to the control group. This result shows that CaMKII activation increase Cpt-1 gene expression in rat skeletal muscle. With respect to mitochondrial integrity, mitochondria size of the exercise group increased by ~3.0 fold compared with the control group, whereas the exercise + KN93 group showed significant decrease compared with the exercise group. Using TEM we show that exercise-induced CaMKII activation increases mitochondria size in rat skeletal muscle and its integrity.Biography
Emmanuel Mukwevho has completed his PhD in 2010 from University of Cape Town, South Africa in Anatomy and Cell Biology. He is an Associate Professor of Biochemistry at North West University, South Africa. He has published both nationally and internationally in reputed journals and his specialty is in Obesity and Diabetes where he led the Diabetes & Obesity Therapeutics Research group at North West University.
Email: emmanuel.mukwevho@nwu.ac.za

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi