Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
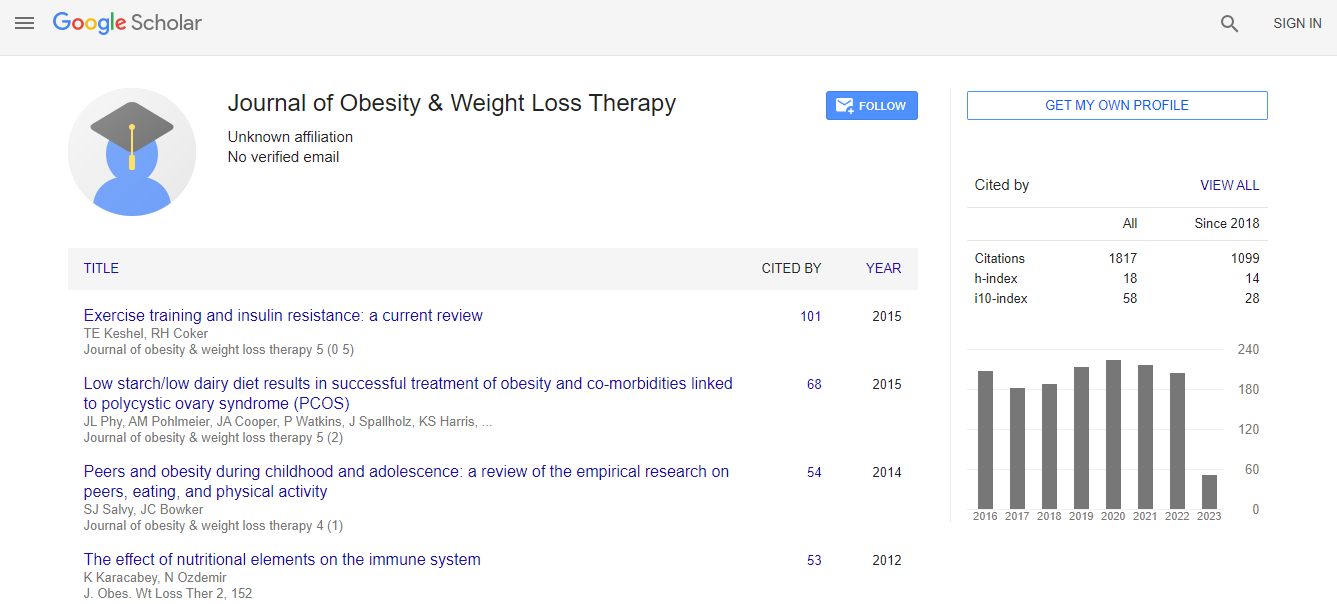
Google Scholar citation report
Citations : 2305
Journal of Obesity & Weight Loss Therapy received 2305 citations as per Google Scholar report
Journal of Obesity & Weight Loss Therapy peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Open J Gate
- Genamics JournalSeek
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- University of Bristol
- Pubmed
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Disease mechanisms and dietary intervention for obesity and T2DM
19th World Obesity Congress
Shaodong Guo
Texas A&M University, USA
ScientificTracks Abstracts: J Obes Weight Loss Ther
Abstract
Insulin resistance serves as the major mechanism for the development of obesity, which is pandemic in population worldwide over the past decades, largely owing to over nutrition. Excess energy stores in the adipose tissue and other organs as lipids, promoting lipotoxicity and metabolic inflammation, activating intracellular protein kinases to impair insulin signaling components and resulting in insulin resistance. Insulin resistance is the key etiologic illness that defines metabolic syndrome, a set of interrelated disorders/issues, inclusive of obesity, hyperglycemia, dyslipidemia and hypertension. Following insulin resistance, a lot of patients with the metabolic syndrome sooner or later developed pancreatic �?²-cell failure, which triggers the onset of Type-2 Diabetes Mellitus (T2DM) and its complications. Our cell and animal-based totally studies demonstrate that insulin and its signaling cascades generally control cell growth, metabolism and survival through activation of Mitogen- Activated Protein Kinases (MAPKs) and Phosphotidylinositide-3-Kinase (PI3K), of which activation of PI-3K-associated with Insulin Receptor Substrate-1 and -2 (IRS 1, 2) and subsequent Aktâ�?�?Fo xo1 phosphorylation cascade has a central function in control of nutrient homeostasis and organ survival. Inactivation of Akt and activation of Foxo1, through suppression IRS1 and IRS2 in a variety of organs following over nutrition, lipotoxicity and inflammation may form a fundamental mechanism for insulin resistance in humans. This seminar discusses the premise of insulin signaling, resistance and how excess nutrients and lipid signaling from obesity promotes infection and insulin resistance, selling organ failure with emphasis on the IRS and the fork head/winged-helix transcription component Foxo1.Biography
Shaodong Guo is an Associate Professor in the Department of Nutrition and Food Science at Texas A&M University College. He has received his PhD in Physiology from Peking University, China. Later, he has completed his Postdoctoral research training in Genetics, Biochemistry and Medicine in the Chinese Academy of Sciences, the University of Illinois at Chicago and Harvard University, respectively. He was an Instructor in Medicine at Children’s Hospital Boston and Harvard Medical School for two years prior to joining the Faculty at Texas A&M Health Science Center. Currently, he serves as a Senior Editor for the Journal of Endocrinology and Journal of Molecular Endocrinology, two major official journals of Endocrine Society of Europe, UK and Australia and he is the author of the textbook chapter for metabolic syndrome and published by Springer in 2016. His lab research focuses on insulin/glucagon and estrogen signal transduction, insulin resistance, gene transcriptional control of nutrient homeostasis and cardiac dysfunction in diabetes. He is a recipient of ADA Junior Faculty Award, Career Development Award and Richard R Lee Award. His work has been published in several journals including the JBC, Endocrinology, Hypertension, Diabetes, Circulation Research, AJP, MCB and Nature Medicine, receiving more than 5,000 citations from the Google Scholar.
E-mail: shaodong.guo@tamu.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi