Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
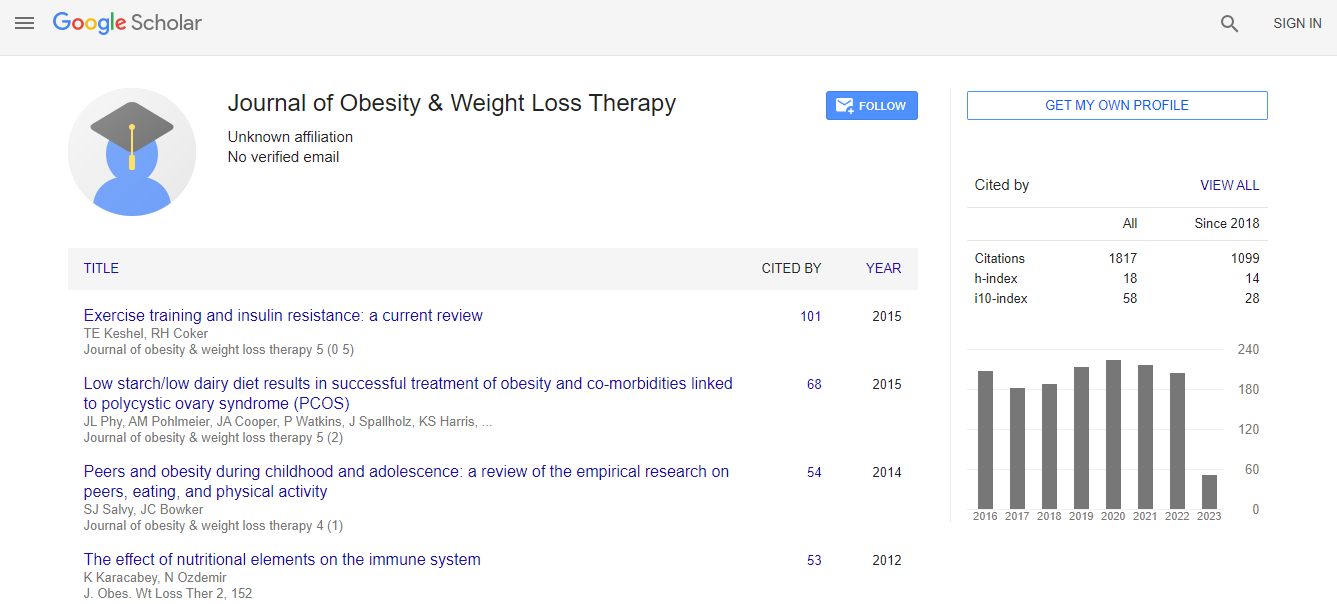
Google Scholar citation report
Citations : 2305
Journal of Obesity & Weight Loss Therapy received 2305 citations as per Google Scholar report
Journal of Obesity & Weight Loss Therapy peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Open J Gate
- Genamics JournalSeek
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- University of Bristol
- Pubmed
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Endocannabinoid control of gut-brain hunger signaling
3rd International Conference and Exhibition on Obesity & Weight Management
Nicholas V Di Patrizio
University of California, USA
ScientificTracks Abstracts-Workshop: J Obes Weight Loss Ther
Abstract
Endocannabinoids are lipid-derived signaling molecules that control food intake and energy metabolism. Recent evidence from our laboratory suggests that tasting dietary fats triggers endocannabinoid signaling within the rat upper small intestine, and this signaling event promotes the intake and possibly preference for fat-rich foods. We now report a broader role for the gut endocannabinoid system in the control of hunger and eating, and identify critical molecular and neural pathways that regulate these physiological processes. Food deprivation induces biosynthesis of the orexigenic endocannabinoid, 2-arachidonoyl-snglycerol (2-AG), in the jejunum mucosa of rats in a time-dependent manner through a mechanism that includes increases in local levels of the 2-AG precursor, 1-stearoyl,2-arachidonoyl-sn-glycerol (SAG). Treatment with tetrahydrolipstatin-an inhibitor of diacylglycerol lipase (DGL), which is a critical enzyme that hydrolyzes SAG and generates 2-AG-inhibits production of jejunal 2-AG, suggesting a role for DGL in the formation of 2-AG in the intestine. Furthermore, deprivation-induced biosynthesis of 2-AG in the jejunum is blocked by complete subdiaphragmaticvagotomy or peripheral treatment with the muscarinic acetylcholine receptor antagonist, atropine. Peripheral administration of atropine, or the peripherally-restricted cannabinoid CB1R antagonist, AM6546, inhibits re-feeding after a 24-hr fast. Collectively, the results suggest that food deprivation drives the biosynthesis of 2-AG in the jejunum through a vagal mechanism that includes activation of local muscarinic acetylcholine receptors. Moreover, this gut-brain signaling pathway might participate more broadly in the integrative and dynamic control of feeding and energy balance.Biography
Nicholas V Di Patrizio’s passion for scientific discovery was solidified during his undergraduate studies at Temple University in Philadelphia. He subsequently earned a Biomedical PhD in the Neurosciences at Drexel University College of Medicine, and did his Postdoctoral research at the University of California, Irvine School of Medicine. He is a recipient of the prestigious National Institutes of Health Pathway to Independence K99/R00 grant award from the National Institute on Drug Abuse. He recently launched his independent research program as an Assistant Professor of Biomedical Sciences at the University of California, Riverside School of Medicine.
Email: ndipatri@uci.edu

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi