Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
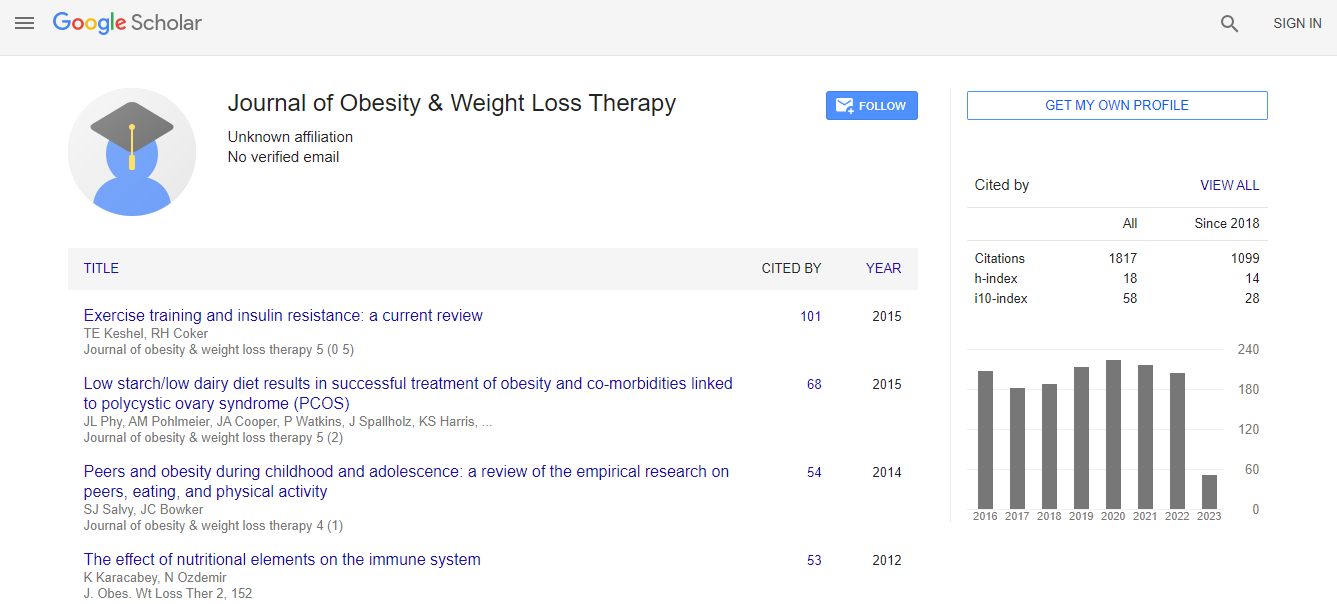
Google Scholar citation report
Citations : 2305
Journal of Obesity & Weight Loss Therapy received 2305 citations as per Google Scholar report
Journal of Obesity & Weight Loss Therapy peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Open J Gate
- Genamics JournalSeek
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- University of Bristol
- Pubmed
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Maternal obesity leads offspring to alterations in miRNA expression and metabolic phenotype and negatively impacts in glucose and lipid homeostasis of F2 generation
JOINT EVENT 10th International Conference on Childhood Obesity and Nutrition & 2nd International Conference on Metabolic and Bariatric Surgery
Lais Simino, Mancini M, Panzarin C, Fontana M F, Milanski M, Torsoni M A, Lgnacio-Souza L M and Torsoni A S
State University of Campinas, Brazil
Posters & Accepted Abstracts: J Obes Weight Loss Ther
Abstract
Background & Aim: Changes in nutritional status in embryonic development and lactation period, as an excessive caloric intake, may lead to a phenomenon known as metabolic programming. Moreover, recent studies have shown that maternal obesity can have transgenerational effects, affecting not only the F1 generation, but also future generations. These effects, transmitted across generations, can be triggered by epigenetics mechanisms, such as miRNA expression. The miRNA Let-7 is shown to be involved in glucose homeostasis and here, we aimed to evaluate its expression in the liver of F1 offspring from obese mothers and the impacts of its modulation to the F2 generation. Methods: Female Swiss mice were fed with a HF or control diet for an adaptation period and through gestation and lactation. Weaned offspring received control diet until d28. Part of female offspring remained in control diet until mating to generate F2 offspring, which were weaned and received control diet until d28. Results: After the adaptation period, F0 females that consumed a HFD were divided in two groups: Obese prone (OP) or obese resistant (OR), according to their weight gain. OP presented higher body weight, adiposity, serum glucose and NEFA than OR. Male and female offspring from OR and OP (OR-O and OP-O) showed an increase in body weight and adiposity at d28, but OP-O presented impaired glucose tolerance and insulin sensitivity, besides higher serum lipid biomarkers. F1 OP-O also had an overexpression in hepatic Let-7 and down-regulation of AMPK, a predicted mRNA target of this miRNA. F2 offspring showed no alteration in body weight and adiposity, but F2 OP-O presented higher fasting glucose as early as d0 and d28, and an elevated liver fat content. Conclusion: Nutritional overload in critical periods of development leads offspring to epigenetic changes that may have transgenerational negative impacts.Biography
Laís Simino is a Nutritionist and pursuing her PhD at State University of Campinas – UNICAMP. She belongs to the Obesity and Comorbidities Research Center (OCRC) and Laboratory of Metabolic Diseases, a laboratory that has been specializing in fetal programming research, especially triggered by maternal consumption of high fat diets.
Email: lais.simino@fca.unicamp.br

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi