Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
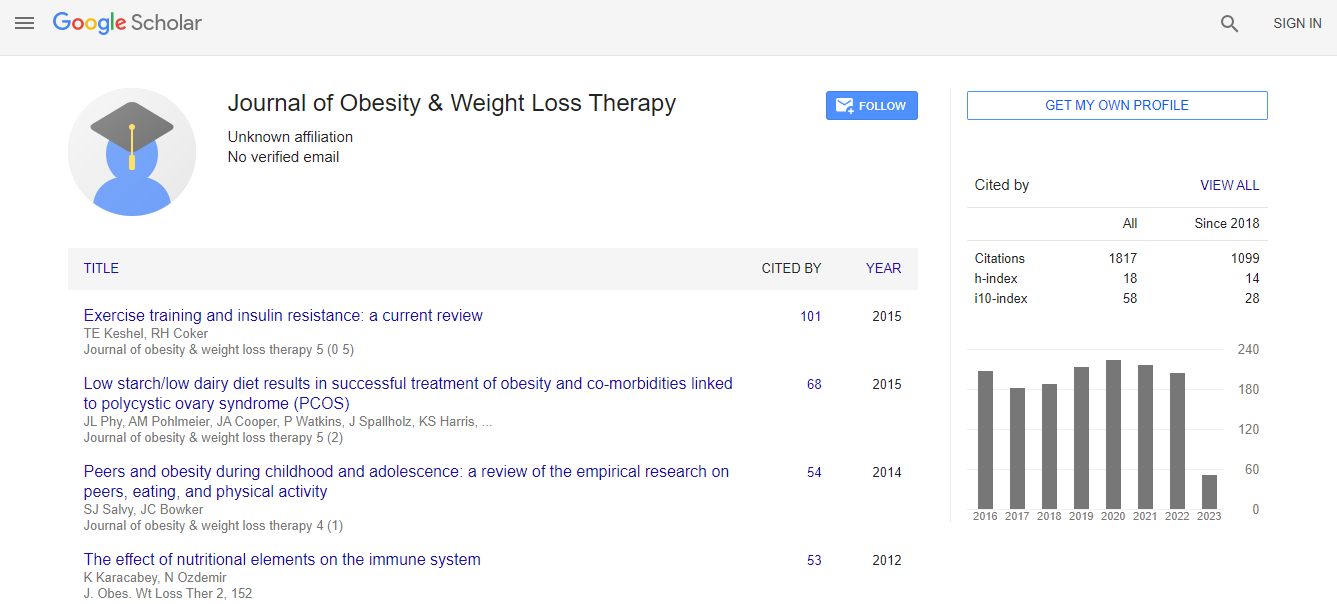
Google Scholar citation report
Citations : 2305
Journal of Obesity & Weight Loss Therapy received 2305 citations as per Google Scholar report
Journal of Obesity & Weight Loss Therapy peer review process verified at publons
Indexed In
- Index Copernicus
- Google Scholar
- Open J Gate
- Genamics JournalSeek
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- CABI full text
- Cab direct
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- University of Bristol
- Pubmed
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Sex differences in the effect of high-fat diet on mouse white adipose tissue
21st World Obesity Conference
Nadezhda Bazhan,Yakovleva T, Dubinina Aand Makarova E
The Institute of Cytology and Genetics,Russia, Federation
Keynote: J Obes Weight Loss Ther
DOI: 10.4172/2165-7904-C10-083
Abstract
Fibroblast growth factor-21 (FGF21) is a circulating hepatokine, favors white adipose tissue (WAT) glucose utilization and lipolysis in mice. Diet induced obesity (DIO) is potentially FGF21-resistant state: Increased circulating FGF21 levels are associated with decreased FGF21 signaling in obese mice. It is unknown whether DIO changes WAT FGF21 signaling and metabolic gene expressions in a sex-specific manner. Obesity was induced by high-calorie diet (10 weeks) in C57Bl mice of both sexes. Blood parameters and visceral WAT expressions of genes involved in FGF21 signaling (FGF21, PPARγ, PGC1α, KLB), glucose input (SLC2A1, SLC2A4), lipolysis (Hsl), lipogenesis (Lpl), fatty acid oxidation (Cpt1), and thermogenesis (Ucp1) were analyzed by RT-PCR. DIO increased WAT indexes and circulating FGF21 levels in mice of both sexes, but WAT content was much higher and FGF21 blood levels were much lower in obese females than in obese males. DIO induced WAT FGF21 gene expression only in male. There were signs of FGF21 resistance (reduced Pgc1α and Slc1a expression) in WAT of obese males. However, increased circulating FGF21 levels and local WAT FGF21 expression seem to ensure adequate expression of other FGF21 target genes and reduced WAT accumulation in obese males as compared to females. There were signs of FGF21 resistance, insulin resistance, and dysregulation of lipid turnover (reduced PPARΓ, SLC2A4, HSL, and LPL expression) in WAT of obese females. These transcription changes in combination with low circulating FGF21 level could lead to pronounce WAT accumulation in obese females. The study was supported by the Russian Science Foundation, Grant No 17-15-01036.Biography
Bazhan N is currently a Chief Researcher at the Institute of Cytology and Genetics, Russian Academy of Sciences and Professor of Novosibirsk State University. She studied on molecular-physiological mechanisms underlying genetic melanocortin obesity, the role of central melanocortin system in the development of stress-induced anorexia; the role of hypothalamo-pituitary-adrenal axis in the food-intake regulation and the mechanisms of metabolic changes associated with age in mice.
E-mail: bazhan-nm@yandex.ru

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi