Our Group organises 3000+ Global Conferenceseries Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
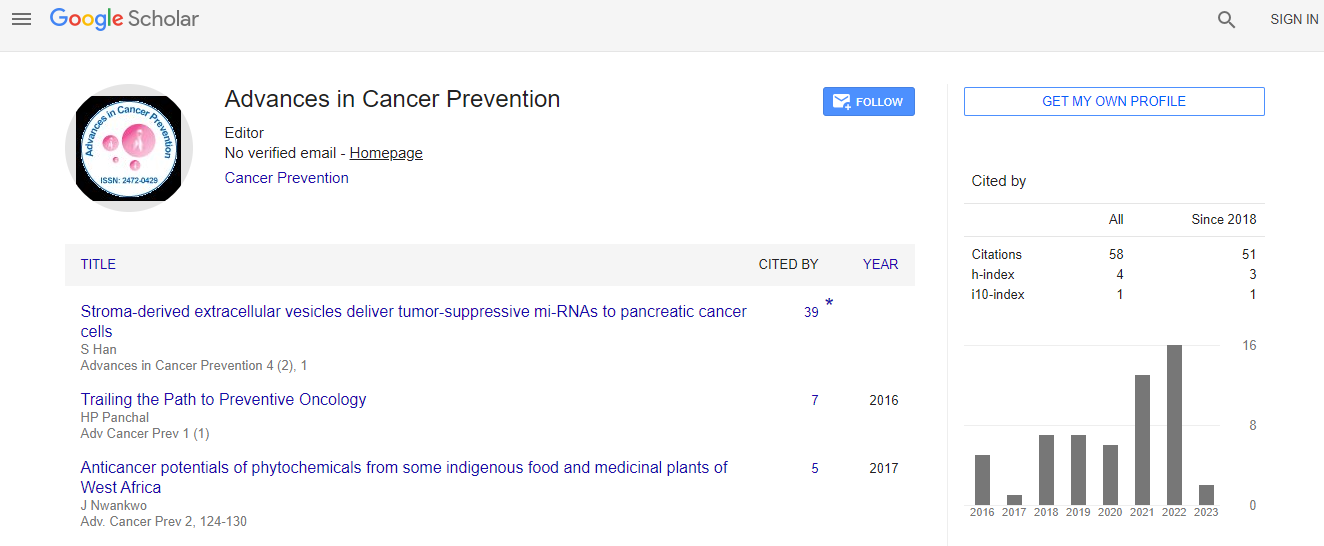
Google Scholar citation report
Citations : 352
Advances in Cancer Prevention received 352 citations as per Google Scholar report
Indexed In
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
Ann Richmond

Ann Richmond
Ingram Professor and Vice Chair
Associate Director of Education
Department of Cancer Biology
Vanderbilt Ingram Cancer Center
USA
Tel: 615-343-7777
Biography
Dr. Richmond received her PhD from Emory University in 1979. She joined Vanderbilt University Dept. of Cell Biology 1989. Currently, she serves as the Ingram Professor of Cancer Biology and Vice Chair of that department. A major question we are trying to answer in my laboratory is "What is the role of the inflammatory response in tumor progression". We are studying the role of proteins that promote the migration of inflammatory cells into tissues. These "chemotactic" proteins can educate leukocytes to either stimulate or inhibit tumor progression. These factors can also stimulate the growth of the tumor and recruit blood vessels into the tumor to provide a continuous supply of nutrients to feed tumor growth. We have tested with a variety of pharmaceutical drugs that shut down the inflammatory process and alter the expression of genes that recruit inflammatory cells into the tumor microenvironment. We are also evaluating how to deliver therapies that teach the patient's leukocytes to fight the growth of the tumor and switch from a pro-tumorigenic to an anti-tumorigenic state. We have also learned that this inflammatory process, combined with other genetic and environmental factors, contributes to mutations in genes that regulate the growth of cells. Some of these mutations make the cells capable of continuous growth and enable the cancer cells to spread throughout the body and grow in distant organs. By determining what genes become mutated or amplified in each patient's tumor and then providing therapies that specifically inhibit the activity of that mutated or amplified gene, we can deliver "personalized cancer treatment" that addresses the problem in that persons specific cancer. This works so much better than the "one size fits all" approach often used previously in chemotherapy. Recently we have determined that inhibition of aurora kinases induce tumor cells to undergo senescence and when we inhibit aurora kinases while at the same time activate the tumor suppressor p53 or the death receptor DR5, melanoma cells die and tumors regress. We are also interested in evaluating how therapeutic agents that target driver mutations such as PI3K, RAS or BRAF affect the tumor microenvironment and the immune response to the tumor. We postulate that in some instances drug resistance comes in part due to deleterious effects of the therapy on the tumor microenvironment. We collaborate with medical oncologists, surgical oncologists, bioengineers, cellular and molecular biologists, and scientists in pharmaceutical companies to address the scientific questions we are asking. This TEAM interaction enables us to optimize our studies to make important breakthroughs in tumor biology and tumor therapy. Our research is funded by the TVHS Department of Veterans Affairs, the National Cancer Institute, and the Department of Defense. I work with a wonderful group of postdoctoral fellows, students, and laboratory scientists who dedicate their lives to providing better treatments for cancer patients.
Research Interest
Inflammation and cancer; the role of chemokines in normal physiology and cancer; signal transduction mechanisms for chemokines; novel therapeutics for treatment of melanoma and Breast Reconstruction Surgery

 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi